Effect of Burosumab Compared With Conventional Therapy on Younger vs Older Children With X-linked Hypophosphatemia
- PMID: 35533340
- PMCID: PMC9282253
- DOI: 10.1210/clinem/dgac296
Effect of Burosumab Compared With Conventional Therapy on Younger vs Older Children With X-linked Hypophosphatemia
Abstract
Context: Younger age at treatment onset with conventional therapy (phosphate salts and active vitamin D; Pi/D) is associated with improved growth and skeletal outcomes in children with X-linked hypophosphatemia (XLH). The effect of age on burosumab efficacy and safety in XLH is unknown.
Objective: This work aimed to explore the efficacy and safety of burosumab vs Pi/D in younger (< 5 years) and older (5-12 years) children with XLH.
Methods: This post hoc analysis of a 64-week, open-label, randomized controlled study took place at 16 academic centers. Sixty-one children aged 1 to 12 years with XLH (younger, n = 26; older, n = 35) participated. Children received burosumab starting at 0.8 mg/kg every 2 weeks (younger, n = 14; older, n = 15) or continued Pi/D individually titrated per recommended guidelines (younger, n = 12; older, n = 20). The main outcome measure included the least squares means difference (LSMD) in Radiographic Global Impression of Change (RGI-C) rickets total score from baseline to week 64.
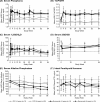
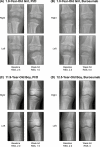
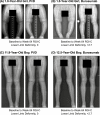
Results: The LSMD in outcomes through 64 weeks on burosumab vs conventional therapy by age group were as follows: RGI-C rickets total score (younger, +0.90; older, +1.07), total Rickets Severity Score (younger, -0.86; older, -1.44), RGI-C lower limb deformity score (younger, +1.02; older, +0.91), recumbent length or standing height Z-score (younger, +0.20; older, +0.09), and serum alkaline phosphatase (ALP) (younger, -31.15% of upper normal limit [ULN]; older, -52.11% of ULN). On burosumab, dental abscesses were not reported in younger children but were in 53% of older children.
Conclusion: Burosumab appears to improve outcomes both in younger and older children with XLH, including rickets, lower limb deformities, growth, and ALP, compared with Pi/D.
Trial registration: ClinicalTrials.gov NCT02915705.
Keywords: X-linked hypophosphatemia; burosumab; children; fibroblast growth factor 23; rickets.
© The Author(s) 2022. Published by Oxford University Press on behalf of the Endocrine Society.
Figures

References
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

