Early remdesivir to prevent severe COVID-19 in recipients of solid organ transplant: a real-life study from Northern Italy
- PMID: 35533831
- PMCID: PMC9076039
- DOI: 10.1016/j.ijid.2022.05.001
Early remdesivir to prevent severe COVID-19 in recipients of solid organ transplant: a real-life study from Northern Italy
Abstract
Objectives: The effectiveness of a 3-day course of remdesivir to prevent severe disease in patients with COVID-19 who received solid organ transplant (SOT) is unknown. We wanted to study the efficacy of this therapeutic option in patients with COVID-19 who received SOT in preventing both hospitalizations for outpatients and clinical worsening due to COVID-19 for those already hospitalized for other reasons.
Methods: This is a single-center, retrospective, observational study conducted in the Fondazione IRCSS Policlinico San Matteo of Pavia, Northern Italy. We extracted all the data of patients with COVID-19 receiving SOT who received and did not receive pre-emptive remdesivir between December 23, 2021, and February 26, 2022. We used a Cox proportional hazard model to assess whether receiving pre-emptive remdesivir was associated with lower rates of hospitalization.
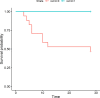
Results: A total of 24 patients who received SOT were identified. Among these, seven patients (29, 1%) received pre-emptive remdesivir, whereas 17 (70, 9%) patients did not. Receiving remdesivir significantly reduced the hospitalization rate in outpatients who received SOT and the clinical worsening of the condition of already hospitalized patients who received SOT (hazard ratio 0.05; confidence interval [0.00-0.65], P-value = 0.01).
Conclusion: In our cohort of patients infected with SARS-CoV-2 who received SOT, pre-emptive remdesivir was effective in reducing the hospitalization rate due to COVID-19 and in preventing the clinical worsening of the condition of patients who received SOT who were hospitalized for reasons other than COVID-19.
Keywords: 3-day course remdesivir; COVID-19; Solid organ transplant recipients.
Copyright © 2022. Published by Elsevier Ltd.
Figures
References
-
- AIFA, AIFA statement. https://farmaci.agenziafarmaco.gov.it/aifa/servlet/PdfDownloadServlet?pd..., 2022. (accessed 4 March 2022).
-
- National Institutes of Health, COVID-19 treatment guidelines: clinical spectrum of SARS-CoV-2 infection. https://www.covid19treatmentguidelines.nih.gov/overview/clinicalspectrum..., 2022a. (accessed 4 March 2022).
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous