F18-FDG PET/CT imaging early predicts pathologic complete response to induction chemoimmunotherapy of locally advanced head and neck cancer: preliminary single-center analysis of the checkrad-cd8 trial
- PMID: 35534690
- PMCID: PMC9226092
- DOI: 10.1007/s12149-022-01744-6
F18-FDG PET/CT imaging early predicts pathologic complete response to induction chemoimmunotherapy of locally advanced head and neck cancer: preliminary single-center analysis of the checkrad-cd8 trial
Abstract
Aim: In the CheckRad-CD8 trial patients with locally advanced head and neck squamous cell cancer are treated with a single cycle of induction chemo-immunotherapy (ICIT). Patients with pathological complete response (pCR) in the re-biopsy enter radioimmunotherapy. Our goal was to study the value of F-18-FDG PET/CT in the prediction of pCR after induction therapy.
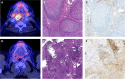
Methods: Patients treated within the CheckRad-CD8 trial that additionally received FDG- PET/CT imaging at the following two time points were included: 3-14 days before (pre-ICIT) and 21-28 days after (post-ICIT) receiving ICIT. Tracer uptake in primary tumors (PT) and suspicious cervical lymph nodes (LN +) was measured using different quantitative parameters on EANM Research Ltd (EARL) accredited PET reconstructions. In addition, mean FDG uptake levels in lymphatic and hematopoietic organs were examined. Percent decrease (Δ) in FDG uptake was calculated for all parameters. Biopsy of the PT post-ICIT acquired after FDG-PET/CT served as reference. The cohort was divided in patients with pCR and residual tumor (ReTu).
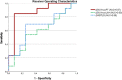
Results: Thirty-one patients were included. In ROC analysis, ΔSUVmax PT performed best (AUC = 0.89) in predicting pCR (n = 17), with a decline of at least 60% (sensitivity, 0.77; specificity, 0.93). Residual SUVmax PT post-ICIT performed best in predicting ReTu (n = 14), at a cutpoint of 6.0 (AUC = 0.91; sensitivity, 0.86; specificity, 0.88). Combining two quantitative parameters (ΔSUVmax ≥ 50% and SUVmax PT post-ICIT ≤ 6.0) conferred a sensitivity of 0.81 and a specificity of 0.93 for determining pCR. Background activity in lymphatic organs or uptake in suspected cervical lymph node metastases lacked significant predictive value.
Conclusion: FDG-PET/CT can identify patients with pCR after ICIT via residual FDG uptake levels in primary tumors and the related changes compared to baseline. FDG-uptake in LN + had no predictive value.
Trial registry: ClinicalTrials.gov identifier: NCT03426657.
Keywords: FDG-PET/CT; HNSCC; Head neck cancer; Immunotherapy; Induction therapy.
© 2022. The Author(s).
Conflict of interest statement
M.B. conflict of interest with Novartis (travel expenses); Bayer (advisory role, honoraria); SIRTEX (travel expenses). J.H. has no conflict of interest. M.E. conflict of interest with Diaceutics (employment, honoraria, advisory role, speakers’ bureau, travel expenses); AstraZeneca (honoraria, advisory role, speakers’ bureau, travel expenses); Roche (honoraria, travel expenses); MSD (honoraria, speakers’ bureau); GenomicHealth (honoraria, advisory role, speakers bureau, travel expenses); Astellas (honoraria, speakers’ bureau); Janssen-Cilag (honoraria, advisory role, research funding, travel expenses); Stratifyer (research funding, patents). D.S. has no conflict of interest. A.O.G. has no conflict of interest. S.M. has no conflict of interest. S.R. conflict of interest with AstraZeneca (research funding); MSD (research funding). U.S.G. conflict of interest with AstraZeneca (advisory role, research funding); BMS (advisory role); MSD (research funding); Sennewald Medizintechnik (travel expenses). J.G. has no conflict of interest. I.T. has no conflict of interest. M.G.H. conflict of interest with Roche (stock, honoraria); Varian (stock); Sanofi (honoraria); AstraZeneca (honoraria, travel expenses); BMS (honoraria, advisory role); MSD (honoraria, advisory role); Merck Serono (honoraria); Celgene (honoraria). G.K. conflict of interest with BMS (advisory role); Lilly (advisory role); Roche (advisory role). J.D. has no conflict of interest. B.T. conflict of interest with BMS (honoraria, advisory role, travel expenses); Merck Serono (honoraria, advisory role); MSD (travel expenses). A.H. conflict of interest with BMS (honoraria, advisory role); MSD (honoraria, advisory role); Roche (honoraria, advisory role, research funding); AstraZeneca (honoraria, advisory role, research funding); Boehringer Ingelheim (honoraria); Abbvie (honoraria); Cepheid (advisory role, research funding); Quiagen (advisory role); Janssen-Cilag (honoraria, advisory role, research funding); Ipsen (honoraria, advisory role); NanoString Technologies (advisory role, research funding, expert testimony); Illumina (advisory role); 3DHistech (advisory role); Diaceutics (advisory role); BioNTech (research funding). H.I. has no conflict of interest. T.K. conflict of interest with Siemens Healthineers (advisory role, honoraria, speakers’ bureau, travel expenses, research funding). R.F. conflict of interest with MSD (honoraria, advisory role, research funding, travel expenses); Fresenius (honoraria); BrainLab (honoraria); AstraZeneca (honoraria, advisory role, research funding, travel expenses); Merck Serono (advisory role, research funding, travel expenses); Novocure (advisory role, speakers’ bureau, research funding); Sennewald (speakers’ bureau, travel expenses). The other authors declare no conflicts of interest. M.H. conflict of interest with Merck Serono (advisory role, speakers’ bureau, honoraria, travel expenses, research funding); MSD (advisory role, speakers’ bureau, travel expenses, research funding); AstraZeneca (research funding); Novartis (research funding); BMS (advisory role, honoraria, speakers’ bureau); Teva (travel expenses). S.S. conflict of interest with Strycker (stock); Varian (stock); Abbot (stock); Crispr Techn. (stock); Pfitzer (stock); Merck Serono (stock); Symrise (stock); Ortho (honoraria, advisory role, speakers’ bureau, research funding, travel expenses); PharmaMar (speakers’ bureau, travel expenses); Haema (speakers’ bureau).
Figures

References
-
- Fietkau R, Hecht M, Hofner B, Lubgan D, Iro H, Gefeller O, et al. Randomized phase-III-trial of concurrent chemoradiation for locally advanced head and neck cancer comparing dose reduced radiotherapy with paclitaxel/cisplatin to standard radiotherapy with fluorouracil/cisplatin: the PacCis-trial. Radiother Oncol. 2020;144:209–217. doi: 10.1016/j.radonc.2020.01.016. - DOI - PubMed
-
- Ang KK, Zhang Q, Rosenthal DI, Nguyen-Tan PF, Sherman EJ, Weber RS, et al. Randomized phase III trial of concurrent accelerated radiation plus cisplatin with or without cetuximab for stage III to IV head and neck carcinoma: RTOG 0522. J Clin Oncol. 2014;32(27):2940–2950. doi: 10.1200/JCO.2013.53.5633. - DOI - PMC - PubMed
-
- Mogadas S, Busch CJ, Pflug C, Hanken H, Krull A, Petersen C, et al. Influence of radiation dose to pharyngeal constrictor muscles on late dysphagia and quality of life in patients with locally advanced oropharyngeal carcinoma. Strahlenther Onkol. 2020;196(6):522–529. doi: 10.1007/s00066-019-01572-0. - DOI - PubMed
-
- Forastiere AA, Zhang Q, Weber RS, Maor MH, Goepfert H, Pajak TF, et al. Long-term results of RTOG 91–11: a comparison of three nonsurgical treatment strategies to preserve the larynx in patients with locally advanced larynx cancer. J Clin Oncol. 2013;31(7):845–852. doi: 10.1200/JCO.2012.43.6097. - DOI - PMC - PubMed
-
- Breheret M, Lubgan D, Haderlein M, Hecht M, Traxdorf M, Schmidt D, et al. Single-cycle induction chemotherapy before chemoradiotherapy or surgery in functionally inoperable head and neck squamous cell carcinoma: 10-year results. Eur Arch Otorhinolaryngol. 2020;277(1):245–254. doi: 10.1007/s00405-019-05665-5. - DOI - PubMed
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

