Transdermal delivery for gene therapy
- PMID: 35538189
- PMCID: PMC9089295
- DOI: 10.1007/s13346-022-01138-1
Transdermal delivery for gene therapy
Abstract
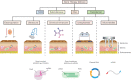
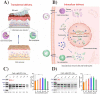
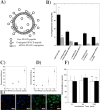
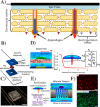
Gene therapy is a critical constituent of treatment approaches for genetic diseases and has gained tremendous attention. Treating and preventing diseases at the genetic level using genetic materials such as DNA or RNAs could be a new avenue in medicine. However, delivering genes is always a challenge as these molecules are sensitive to various enzymes inside the body, often produce systemic toxicity, and suffer from off-targeting problems. In this regard, transdermal delivery has emerged as an appealing approach to enable a high efficiency and low toxicity of genetic medicines. This review systematically summarizes outstanding transdermal gene delivery methods for applications in skin cancer treatment, vaccination, wound healing, and other therapies.
Keywords: DNA; Gene therapy; Microneedles; Transdermal delivery; mRNA; miRNA; siRNA.
© 2022. Controlled Release Society.
Conflict of interest statement
TDN has a conflict of interest with PiezoBioMembrane Inc. and SingleTimeMicroneedles Inc.
Figures





References
-
- Dunbar CE, High KA, Joung JK, Kohn DB, Ozawa K, Sadelain M. Gene therapy comes of age. Science. 2018;359(6372). 10.1126/science.aan4672. - PubMed
-
- FDA. What is gene therapy. 2019 a.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

