Oxidative functionalization of aliphatic and aromatic amino acid derivatives with H2O2 catalyzed by a nonheme imine based iron complex
- PMID: 35539690
- PMCID: PMC9080596
- DOI: 10.1039/c8ra02879f
Oxidative functionalization of aliphatic and aromatic amino acid derivatives with H2O2 catalyzed by a nonheme imine based iron complex
Abstract
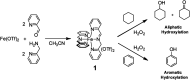
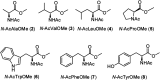
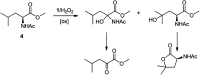
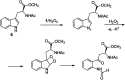
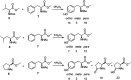
The oxidation of a series of N-acetyl amino acid methyl esters with H2O2 catalyzed by a very simple iminopyridine iron(ii) complex 1 easily obtainable in situ by self-assembly of 2-picolylaldehyde, 2-picolylamine, and Fe(OTf)2 was investigated. Oxidation of protected aliphatic amino acids occurs at the α-C-H bond exclusively (N-AcAlaOMe) or in competition with the side-chain functionalization (N-AcValOMe and N-AcLeuOMe). N-AcProOMe is smoothly and cleanly oxidized with high regioselectivity affording exclusively C-5 oxidation products. Remarkably, complex 1 is also able to catalyze the oxidation of the aromatic N-AcPheOMe. A marked preference for the aromatic ring hydroxylation over Cα-H and benzylic C-H oxidation was observed, leading to the clean formation of tyrosine and its phenolic isomers.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures
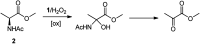
References
-
- Schepartz A. Cuenoud B. J. Am. Chem. Soc. 1990;112:3247–3249. doi: 10.1021/ja00164a075. - DOI
- Hoyer D. Cho H. Schultz P. G. J. Am. Chem. Soc. 1990;112:3249–3250. doi: 10.1021/ja00164a076. - DOI
- Zhong M. Lin L. Kallenbach N. R. Proc. Natl. Acad. Sci. U. S. A. 1995;92:2111–2115. doi: 10.1073/pnas.92.6.2111. - DOI - PMC - PubMed
- Gallagher J. Zelenko O. Walts A. D. Sigman D. S. Biochemistry. 1998;37:2096–2104. doi: 10.1021/bi971565j. - DOI - PubMed
- Xu G. Chance M. R. Chem. Rev. 2007;107:3514–3543. doi: 10.1021/cr0682047. - DOI - PubMed
- Maleknia S. D. Downard K. M. Chem. Soc. Rev. 2014;43:3244–3258. doi: 10.1039/C3CS60432B. - DOI - PubMed
-
- Reddy B. V. S. Reddy L. R. Corey E. J. Org. Lett. 2006;8:3391–3394. doi: 10.1021/ol061389j. - DOI - PubMed
- Annese C. Fanizza I. Calvano C. D. D'Accolti L. Fusco C. Curci R. Williard P. G. Org. Lett. 2011;13:5096–5099. doi: 10.1021/ol201971v. - DOI - PMC - PubMed
- Noisier A. F. M. Brimble M. A. Chem. Rev. 2014;114:8775–8806. doi: 10.1021/cr500200x. - DOI - PubMed
- Gong W. Zhang G. Liu T. Giri R. Yu J.-Q. J. Am. Chem. Soc. 2014;136:16940–16946. doi: 10.1021/ja510233h. - DOI - PMC - PubMed
- Osberger T. J. Rogness D. C. Kohrt J. T. Stepan A. F. White M. C. Nature. 2016;537:214–219. doi: 10.1038/nature18941. - DOI - PMC - PubMed
-
- Easton C. J. Pure Appl. Chem. 1997;69:489–494. doi: 10.1351/pac199769030489. - DOI
-
- Berlett B. S. Stadtman E. R. J. Biol. Chem. 1997;272:20313–20316. doi: 10.1074/jbc.272.33.20313. - DOI - PubMed
- Hawkins C. L. Davies M. J. J. Chem. Soc., Perkin Trans. 2. 1998:2617–2622. doi: 10.1039/A806666C. - DOI
- Saladino R. Mezzetti M. Mincione E. Torrini I. Paglialunga Paradisi M. Mastropietro G. J. Org. Chem. 1999;64:8468–8474. doi: 10.1021/jo990185w. - DOI
- Goshe M. B. Chen Y. H. Anderson V. E. Biochemistry. 2000;39:1761–1770. doi: 10.1021/bi991569j. - DOI - PubMed
- Hawkins C. L. Davies M. J. Biochim. Biophys. Acta. 2001;1504:196–219. doi: 10.1016/S0005-2728(00)00252-8. - DOI - PubMed
- Nukuna B. N. Goshe M. B. Anderson V. E. J. Am. Chem. Soc. 2001;123:1208–1214. doi: 10.1021/ja003342d. - DOI - PubMed
- Dangel B. D. Johnson J. A. Sames D. J. Am. Chem. Soc. 2001;123:8149–8150. doi: 10.1021/ja016280f. - DOI - PubMed
- Croft A. K. Easton C. J. Radom L. J. Am. Chem. Soc. 2003;125:4119–4124. doi: 10.1021/ja029674v. - DOI - PubMed
- Davies M. J. Biochim. Biophys. Acta. 2005;1703:93–109. doi: 10.1016/j.bbapap.2004.08.007. - DOI - PubMed
- Rella M. R. Williard P. G. J. Org. Chem. 2007;72:525–531. doi: 10.1021/jo061910n. - DOI - PMC - PubMed
- Watts Z. I. Easton C. J. J. Am. Chem. Soc. 2009;131:11323–11325. doi: 10.1021/ja9027583. - DOI - PubMed
- Raffy Q. Buisson D.-A. Cintrat J.-C. Rousseau B. Pin S. Renault J. P. Angew. Chem., Int. Ed. 2012;51:2960–2963. doi: 10.1002/anie.201108856. - DOI - PubMed
- Morgan P. E. Pattison D. I. Davies M. J. Free Radical Biol. Med. 2012;52:328–339. doi: 10.1016/j.freeradbiomed.2011.10.448. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

