Vortex-assisted low density solvent and surfactant based dispersive liquid-liquid microextraction for sensitive spectrophotometric determination of cobalt
- PMID: 35540320
- PMCID: PMC9078415
- DOI: 10.1039/c7ra11896a
Vortex-assisted low density solvent and surfactant based dispersive liquid-liquid microextraction for sensitive spectrophotometric determination of cobalt
Abstract
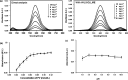
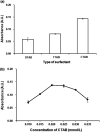
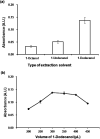
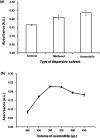
This study describes the development of vortex-assisted low density solvent and surfactant based dispersive liquid-liquid microextraction (VALS-DLLME) for Co(ii) prior to its spectrophotometric detection. The method consisted of the complexation of Co(ii) with pyrocatechol violet (PV) followed by the preconcentration of the Co(II)-PV complex using VALS-DLLME and then an absorption measurement at 600 nm. The optimum conditions for complex formation were a 1 : 3 mole ratio of Co(ii) and PV at pH 7.5, while the conditions for VALS-DLLME were 300 μL 1-dodecanol as extraction solvent, and 300 μL acetonitrile as dispersive solvent under a vortex for 20 s with the addition of cationic surfactant (0.02 mmol L-1 CTAB). Under the optimum conditions, good linearity was in the range of 0.1-10 mg L-1, the enrichment factor (EF) was 13.5 and the low limit of detection (LOD) was 0.04 mg L-1. The method was applied to the analysis of Co(ii) in water, green leaf vegetable and vitamin B12 samples. The proposed method provided good recoveries in the range of 86-104%, which were comparable to those obtained from flame atomic absorption spectrophotometry.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures
Similar articles
-
Green aspects, developments and perspectives of liquid phase microextraction techniques.Talanta. 2014 Feb;119:34-45. doi: 10.1016/j.talanta.2013.10.050. Epub 2013 Oct 29. Talanta. 2014. PMID: 24401382 Review.
-
[Simultaneous determination of polycyclic aromatic hydrocarbons and phthalate esters in surface water by dispersive liquid-liquid microextraction based on solidification of floating organic drop followed by high performance liquid chromatography].Se Pu. 2020 Nov 8;38(11):1308-1315. doi: 10.3724/SP.J.1123.2020.01020. Se Pu. 2020. PMID: 34213102 Chinese.
-
Vortex-assisted low density solvent based demulsified dispersive liquid-liquid microextraction and high-performance liquid chromatography for the determination of organophosphorus pesticides in water samples.Chemosphere. 2014 May;103:51-8. doi: 10.1016/j.chemosphere.2013.11.024. Epub 2013 Dec 13. Chemosphere. 2014. PMID: 24332733
-
Supramolecular solvent-based-vortex-assisted-dispersive liquid liquid microextraction of Sudan Black B in food samples prior to spectrophotometric detection.Turk J Chem. 2024 Jan 2;48(2):329-337. doi: 10.55730/1300-0527.3661. eCollection 2024. Turk J Chem. 2024. PMID: 39050501 Free PMC article.
-
Beyond dispersive liquid-liquid microextraction.J Chromatogr A. 2014 Mar 28;1335:2-14. doi: 10.1016/j.chroma.2014.02.021. Epub 2014 Feb 15. J Chromatogr A. 2014. PMID: 24582396 Review.
Cited by
-
Surface-Active Ionic Liquid-Assisted Cloud Point Extraction for Pre-Concentration and Determination of Cobalt Ions in Pharmaceutical Preparations.Iran J Pharm Res. 2022 May 17;21(1):e127043. doi: 10.5812/ijpr-127043. eCollection 2022 Dec. Iran J Pharm Res. 2022. PMID: 35937559 Free PMC article.
-
A Simple in Syringe Low Density Solvent-Dispersive Liquid Liquid Microextraction for Enrichment of Some Metal Ions Prior to Their Determination by High Performance Liquid Chromatography in Food Samples.Molecules. 2020 Jan 28;25(3):552. doi: 10.3390/molecules25030552. Molecules. 2020. PMID: 32012808 Free PMC article.
-
A Vortex-Assisted Dispersive Liquid-Liquid Microextraction Followed by UPLC-MS/MS for Simultaneous Determination of Pesticides and Aflatoxins in Herbal Tea.Molecules. 2019 Mar 15;24(6):1029. doi: 10.3390/molecules24061029. Molecules. 2019. PMID: 30875921 Free PMC article.
References
LinkOut - more resources
Full Text Sources

