Quinoline-triazole hybrids inhibit falcipain-2 and arrest the development of Plasmodium falciparum at the trophozoite stage
- PMID: 35540629
- PMCID: PMC9076119
- DOI: 10.1039/c9ra06571g
Quinoline-triazole hybrids inhibit falcipain-2 and arrest the development of Plasmodium falciparum at the trophozoite stage
Abstract
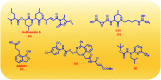
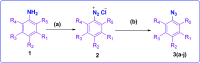
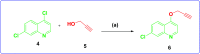
Falcipain-2 (FP2) is a papain family cysteine protease and a key member of the hemoglobin degradation pathway, a process that is required at erythrocytic stages of Plasmodium falciparum to obtain amino acids. In this study, we report a set of 10 quinoline-triazole-based compounds (T1-T10) which exhibit a good binding affinity for FP2, inhibit its catalytic activity at micromolar concentrations and thereby arrest the parasite growth. Compounds T4 and T7 inhibited FP2 with IC50 values of 16.16 μM and 25.64 μM respectively. Both the compounds T4 and T7 arrested the development of P. falciparum at the trophozoite stage with an EC50 value 21.89 μM and 49.88 μM. These compounds also showed morphological and food-vacuole abnormalities like E-64, a known inhibitor of FP2. Our results thus identify the quinoline-triazole-based compounds as a probable starting point for the design of FP2 inhibitors and they should be further investigated as potential antimalarial agents.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures



Similar articles
-
Quinoline carboxamide core moiety-based compounds inhibit P. falciparumfalcipain-2: Design, synthesis and antimalarial efficacy studies.Bioorg Chem. 2021 Mar;108:104514. doi: 10.1016/j.bioorg.2020.104514. Epub 2020 Nov 24. Bioorg Chem. 2021. PMID: 33280833
-
Inhibition of Hemoglobin Degrading Protease Falcipain-2 as a Mechanism for Anti-Malarial Activity of Triazole-Amino Acid Hybrids.Curr Top Med Chem. 2020;20(5):377-389. doi: 10.2174/1568026620666200130162347. Curr Top Med Chem. 2020. PMID: 32000644
-
Inhibition of Plasmodium falciparum cysteine protease falcipain-2 by a human cross-class inhibitor serpinB3: A mechanistic insight.Biochim Biophys Acta Proteins Proteom. 2019 Sep;1867(9):854-865. doi: 10.1016/j.bbapap.2019.06.012. Epub 2019 Jun 24. Biochim Biophys Acta Proteins Proteom. 2019. PMID: 31247344
-
Falcipain cysteine proteases of malaria parasites: An update.Biochim Biophys Acta Proteins Proteom. 2020 Mar;1868(3):140362. doi: 10.1016/j.bbapap.2020.140362. Epub 2020 Jan 9. Biochim Biophys Acta Proteins Proteom. 2020. PMID: 31927030 Review.
-
Cysteine proteases of malaria parasites.Int J Parasitol. 2004 Dec;34(13-14):1489-99. doi: 10.1016/j.ijpara.2004.10.003. Int J Parasitol. 2004. PMID: 15582526 Review.
Cited by
-
Is structural hybridization invoking new dimensions for antimalarial drug discovery research?RSC Med Chem. 2023 May 4;14(7):1227-1253. doi: 10.1039/d3md00083d. eCollection 2023 Jul 20. RSC Med Chem. 2023. PMID: 37484560 Free PMC article. Review.
-
Fabrication of copper(II)-coated magnetic core-shell nanoparticles Fe3O4@SiO2-2-aminobenzohydrazide and investigation of its catalytic application in the synthesis of 1,2,3-triazole compounds.Sci Rep. 2021 Jan 22;11(1):2073. doi: 10.1038/s41598-021-81632-7. Sci Rep. 2021. PMID: 33483570 Free PMC article.
-
Synthesis of new series of quinoline derivatives with insecticidal effects on larval vectors of malaria and dengue diseases.Sci Rep. 2022 Mar 19;12(1):4765. doi: 10.1038/s41598-022-08397-5. Sci Rep. 2022. PMID: 35306526 Free PMC article.
-
Reinvestigation of diphenylmethylpiperazine analogues of pyrazine as new class of Plasmodial cysteine protease inhibitors for the treatment of malaria.RSC Med Chem. 2024 Feb 1;15(3):1022-1037. doi: 10.1039/d3md00490b. eCollection 2024 Mar 20. RSC Med Chem. 2024. PMID: 38516592 Free PMC article.
-
Three Decades of Targeting Falcipains to Develop Antiplasmodial Agents: What have we Learned and What can be Done Next?Curr Med Chem. 2024;31(16):2234-2263. doi: 10.2174/0929867331666230913165219. Curr Med Chem. 2024. PMID: 37711130 Review.
References
LinkOut - more resources
Full Text Sources
Research Materials

