Plasmon-activated water effectively relieves hepatic oxidative damage resulting from chronic sleep deprivation
- PMID: 35540828
- PMCID: PMC9078654
- DOI: 10.1039/c7ra13559a
Plasmon-activated water effectively relieves hepatic oxidative damage resulting from chronic sleep deprivation
Abstract
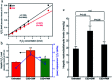
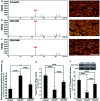
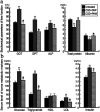
The role of the hepato-protective agent plasmon-activated water (PAW) as an innovative anti-oxidant during chronic sleep deprivation (SD) is realized in this study. PAW possesses reduced hydrogen-bonded structure, higher chemical potential and significant anti-oxidative properties. In vitro tests using rat liver cell line (Clone-9) have demonstrated that PAW is non-cytotoxic and does not change the cellular migration capacity. The in vivo experiment on SD rats suffering from intense oxidative damage to the liver, an extremely common phenomenon in the present-time with deleterious effects on metabolic function, is performed by feeding PAW to replace deionized (DI) water. Experimental results indicate that PAW markedly reduces oxidative stress with enhanced bioenergetics in hepatocytes. PAW also effectively restores hepatocytic trans-membrane ion homeostasis, preserves membranous structures, and successfully improves liver function and metabolic activity. In addition, the hepato-protective effects of PAW are evidently demonstrated by the reduced values of glutamic oxaloacetic transaminase (GOT) and glutamic pyruvic transaminase (GPT) and the recovery of total protein and albumin levels. With clear evidences of PAW for protecting liver from SD-induced injury, delivering PAW as a powerful hepato-protective agent should be worthy of trailblazing new clinical trials in a healthier, more natural, and more convenient way.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures




Similar articles
-
Plasmon-Activated Water Reduces Amyloid Burden and Improves Memory in Animals with Alzheimer's Disease.Sci Rep. 2019 Sep 13;9(1):13252. doi: 10.1038/s41598-019-49731-8. Sci Rep. 2019. PMID: 31520077 Free PMC article.
-
Influence of ferulic acid consumption in ameliorating the cadmium-induced liver and renal oxidative damage in rats.Environ Sci Pollut Res Int. 2019 Jul;26(20):20631-20653. doi: 10.1007/s11356-019-05420-7. Epub 2019 May 18. Environ Sci Pollut Res Int. 2019. PMID: 31104231
-
Transferring Plasmon Effect on a Biological System: Expression of Biological Polymers in Chronic Rejection and Inflammatory Rat Model.Polymers (Basel). 2021 May 31;13(11):1827. doi: 10.3390/polym13111827. Polymers (Basel). 2021. PMID: 34072966 Free PMC article.
-
Sleep deprivation-induced multi-organ injury: role of oxidative stress and inflammation.EXCLI J. 2015 May 18;14:672-83. doi: 10.17179/excli2015-245. eCollection 2015. EXCLI J. 2015. PMID: 26648820 Free PMC article.
-
Cooperation of liver cells in health and disease.Adv Anat Embryol Cell Biol. 2001;161:III-XIII, 1-151. doi: 10.1007/978-3-642-56553-3. Adv Anat Embryol Cell Biol. 2001. PMID: 11729749 Review.
Cited by
-
Plasmon-activated water enhances gut-barrier function and alleviates inflammation in a mouse model of ulcerative colitis.Exp Ther Med. 2025 May 27;30(1):146. doi: 10.3892/etm.2025.12896. eCollection 2025 Jul. Exp Ther Med. 2025. PMID: 40496763 Free PMC article.
-
Combined Lycium barbarum Polysaccharides with Plasmon-Activated Water Affect IFN-γ/TNF-α Induced Inflammation in Caco-2 Cells.Pharmaceuticals (Basel). 2023 Oct 13;16(10):1455. doi: 10.3390/ph16101455. Pharmaceuticals (Basel). 2023. PMID: 37895926 Free PMC article.
-
Plasmon-Activated Water can Prolong Existing Sea-Ice Habitats to Potentially Save Polar Bears.Sci Rep. 2019 Jul 18;9(1):10398. doi: 10.1038/s41598-019-46867-5. Sci Rep. 2019. PMID: 31320695 Free PMC article.
-
Therapeutics for Inflammatory-Related Diseases Based on Plasmon-Activated Water: A Review.Int J Mol Sci. 2018 May 28;19(6):1589. doi: 10.3390/ijms19061589. Int J Mol Sci. 2018. PMID: 29843406 Free PMC article. Review.
-
Light, Water, and Melatonin: The Synergistic Regulation of Phase Separation in Dementia.Int J Mol Sci. 2023 Mar 19;24(6):5835. doi: 10.3390/ijms24065835. Int J Mol Sci. 2023. PMID: 36982909 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources

