Single-cell transcriptomics identifies premature aging features of TERC-deficient mouse brain and bone marrow
- PMID: 35545739
- PMCID: PMC9616978
- DOI: 10.1007/s11357-022-00578-4
Single-cell transcriptomics identifies premature aging features of TERC-deficient mouse brain and bone marrow
Abstract
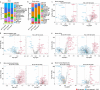
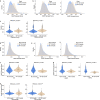
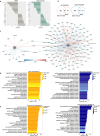
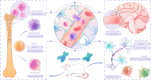
Aging is a progressive loss of physiological function and increased susceptibility to major pathologies. Degenerative diseases in both brain and bone including Alzheimer disease (AD) and osteoporosis are common in aging groups. TERC is RNA component of telomerase, and its deficiency accelerates aging-related phenotypes including impaired life span, organ failure, bone loss, and brain dysfunction. In this study, we investigated the traits of bone marrow-brain cross-tissue communications in young mice, natural aging mice, and premature aging (TERC deficient, TERC-KO) mice by single-cell transcriptome sequencing. Differentially expressed gene analysis of brain as well as bone marrow between premature aging mouse and young mouse demonstrated aging-related inflammatory response and suppression of neuron development. Further analysis of senescence-associated secretory phenotype (SASP) landscape indicated that TERC-KO perturbation was enriched in oligodendrocyte progenitor cells (OPCs) and hematopoietic stem and progenitor cells (HSPC). Series of inflammatory associated myeloid cells was activated in premature aging mice brain and bone marrow. Cross-tissue comparison of TERC-KO mice brain and bone marrow illustrated obvious ligand-receptor communications between brain glia cells, macrophages, and bone marrow myeloid cells in premature aging-induced inflammation. Enrichment of co-regulation modules between brain and bone marrow identified premature aging response genes such as Dusp1 and Ifitm3. Our study provides a rich resource for understanding premature aging-associated perturbation in brain and bone marrow and supporting myeloid cells and endothelial cells as promising therapy targeting for age-related brain-bone diseases.
Keywords: Aging; Brain-bone axis; Single-cell RNA sequencing; TERC; Telomerase.
© 2022. The Author(s), under exclusive licence to American Aging Association.
Conflict of interest statement
The authors declare no competing interests.
Figures



Similar articles
-
Mouse models of telomere dysfunction phenocopy skeletal changes found in human age-related osteoporosis.Dis Model Mech. 2014 May;7(5):583-92. doi: 10.1242/dmm.014928. Epub 2014 Mar 13. Dis Model Mech. 2014. PMID: 24626990 Free PMC article.
-
Telomerase-deficient mice exhibit bone loss owing to defects in osteoblasts and increased osteoclastogenesis by inflammatory microenvironment.J Bone Miner Res. 2011 Jul;26(7):1494-505. doi: 10.1002/jbmr.349. J Bone Miner Res. 2011. PMID: 21308778
-
Hematopoietic lineage skewing and intestinal epithelia degeneration in aged mice with telomerase RNA component deletion.Exp Gerontol. 2015 Dec;72:251-60. doi: 10.1016/j.exger.2015.10.016. Epub 2015 Oct 30. Exp Gerontol. 2015. PMID: 26523501 Free PMC article.
-
Dyskeratosis congenita: molecular insights into telomerase function, ageing and cancer.Expert Rev Mol Med. 2004 Dec 20;6(26):1-23. doi: 10.1017/S1462399404008671. Expert Rev Mol Med. 2004. PMID: 15613268 Review.
-
Glucose Metabolism and Aging of Hematopoietic Stem and Progenitor Cells.Int J Mol Sci. 2022 Mar 11;23(6):3028. doi: 10.3390/ijms23063028. Int J Mol Sci. 2022. PMID: 35328449 Free PMC article. Review.
Cited by
-
Insights into the Pathobiology of GM1 Gangliosidosis from Single-Nucleus Transcriptomic Analysis of CNS Cells in a Mouse Model.Int J Mol Sci. 2024 Sep 8;25(17):9712. doi: 10.3390/ijms25179712. Int J Mol Sci. 2024. PMID: 39273659 Free PMC article.
-
Single-Cell RNA-Seq Reveals the Pseudo-temporal Dynamic Evolution Characteristics of ADSCs to Neuronal Differentiation.Cell Mol Neurobiol. 2024 Dec 11;45(1):5. doi: 10.1007/s10571-024-01524-y. Cell Mol Neurobiol. 2024. PMID: 39661257 Free PMC article.
-
Noncoding RNA Terc-53 and hyaluronan receptor Hmmr regulate aging in mice.Protein Cell. 2025 Jan 4;16(1):28-48. doi: 10.1093/procel/pwae023. Protein Cell. 2025. PMID: 38721690 Free PMC article.
-
Senescence-induced p21high macrophages contributed to CD8+ T cells-related immune hyporesponsiveness in kidney transplantation via Zfp36/IL-27 axis.Cell Discov. 2025 Apr 15;11(1):38. doi: 10.1038/s41421-025-00784-2. Cell Discov. 2025. PMID: 40234384 Free PMC article.
-
Deep coverage and quantification of the bone proteome provides enhanced opportunities for new discoveries in skeletal biology and disease.PLoS One. 2023 Oct 10;18(10):e0292268. doi: 10.1371/journal.pone.0292268. eCollection 2023. PLoS One. 2023. PMID: 37816044 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

