Performance of a Multianalyte 'Rule-Out' Assay in Pregnant Individuals With Suspected Preeclampsia
- PMID: 35545947
- PMCID: PMC9172903
- DOI: 10.1161/HYPERTENSIONAHA.122.19038
Performance of a Multianalyte 'Rule-Out' Assay in Pregnant Individuals With Suspected Preeclampsia
Abstract
Background: The ability to diagnose preeclampsia clinically is suboptimal. Our objective was to validate a novel multianalyte assay and characterize its performance, when intended for use as an aid to rule-out preeclampsia.
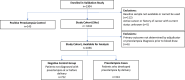
Methods: Prospective, multicenter cohort study of pregnant individuals presenting between 280/7 and 366/7 weeks' with preeclampsia-associated signs and symptoms. Individuals not diagnosed with preeclampsia after baseline evaluation were enrolled in the study cohort, with those who later developed preeclampsia, classified as cases and compared with a negative control group who did not develop preeclampsia. Individuals with assay values at time of enrollment ≥0.0325, determined using a previously developed algorithm, considered at risk. The primary analysis was the time to develop preeclampsia assessed using a multivariate Cox regression model.
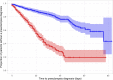
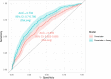
Results: One thousand thirty-six pregnant individuals were enrolled in the study cohort with an incidence of preeclampsia of 30.3% (27.6%-33.2%). The time to develop preeclampsia was shorter for those with an at-risk compared with negative assay result (log-rank P<0.0001; adjusted hazard ratio of 4.81 [3.69-6.27, P<0.0001]). The performance metrics for the assay to rule-out preeclampsia within 7 days of enrollment showed a sensitivity 76.4% (67.5%-83.5%), negative predictive value 95.0% (92.8%-96.6%), and negative likelihood ratio 0.46 (0.32-0.65). Assay performance improved if delivery occurred <37 weeks and for individuals enrolled between 28 and 35 weeks.
Conclusions: We confirmed that a novel multianalyte assay was associated with the time to develop preeclampsia and has a moderate sensitivity and negative likelihood ratio but high negative predictive value when assessed as an aid to rule out preeclampsia within 7 days of enrollment.
Registration: The study was registered on Clinicaltrials.gov (Identifier NCT02780414).
Keywords: biomarkers; hypertension; preeclampsia.
Figures
References
-
- Gestational hypertension and preeclampsia: ACOG practice bulletin, number 222. Obstet Gynecol. 2020;135:e237–e260. doi: 10.1097/AOG.0000000000003891 - PubMed
-
- Firoz T, Sanghvi H, Merialdi M, von Dadelszen P. Pre-eclampsia in low and middle income countries. Best Pract Res Clin Obstet Gynaecol. 2011;25:537–548. doi: 10.1016/j.bpobgyn.2011.04.002 - PubMed
-
- Say L, Chou D, Gemmill A, Tunçalp Ö, Moller AB, Daniels J, Gülmezoglu AM, Temmerman M, Alkema L. Global causes of maternal death: a WHO systematic analysis. Lancet Glob Health. 2014;2:e323–e333. doi: 10.1016/S2214-109X(14)70227-X - PubMed
-
- MacKay AP, Berg CJ, Atrash HK. Pregnancy-related mortality from preeclampsia and eclampsia. Obstet Gynecol. 2001;97:533–538. doi: 10.1016/s0029-7844(00)01223-0 - PubMed
-
- Hoyert DL, Miniño AM. Maternal mortality in the united states: changes in coding, publication, and data release, 2018. Natl Vital Stat Rep. 2020;69:1–18. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

