In vitro and in vivo evaluation of DC-targeting PLGA nanoparticles encapsulating heparanase CD4+ and CD8+ T-cell epitopes for cancer immunotherapy
- PMID: 35546204
- PMCID: PMC10991543
- DOI: 10.1007/s00262-022-03209-1
In vitro and in vivo evaluation of DC-targeting PLGA nanoparticles encapsulating heparanase CD4+ and CD8+ T-cell epitopes for cancer immunotherapy
Abstract
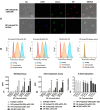
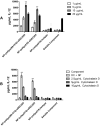
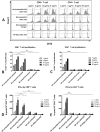
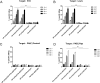
Heparanase has been identified as a universal tumor-associated antigen, but heparanase epitope peptides are difficult to recognize. Therefore, it is necessary to explore novel strategies to ensure efficient delivery to antigen-presenting cells. Here, we established a novel immunotherapy model targeting antigens to dendritic cell (DC) receptors using a combination of heparanase CD4+ and CD8+ T-cell epitope peptides to achieve an efficient cytotoxic T-cell response, which was associated with strong activation of DCs. First, pegylated poly(lactic-coglycolic acid) (PLGA) nanoparticles (NPs) were used to encapsulate a combined heparanase CD4+ and CD8+ T-cell epitope alone or in combination with Toll-like receptor 3 and 7 ligands as a model antigen to enhance immunogenicity. The ligands were then targeted to DC cell-surface molecules using a DEC-205 antibody. The binding and internalization of these PLGA NPs and the activation of DCs, the T-cell response and the tumor-killing effect were assessed. The results showed that PLGA NPs encapsulating epitope peptides (mHpa399 + mHpa519) could be targeted to and internalized by DCs more efficiently, stimulating higher levels of IL-12 production, T-cell proliferation and IFN-γ production by T cells in vitro. Moreover, vaccination with DEC-205-targeted PLGA NPs encapsulating combined epitope peptides exhibited higher tumor-killing efficacy both in vitro and in vivo. In conclusion, delivery of PLGA NP vaccines targeting DEC-205 based on heparanase CD4+ and CD8+ T-cell epitopes are suitable immunogens for antitumor immunotherapy and have promising potential for clinical applications.
Keywords: Dendritic cells; Heparanase; Nanoparticles; Targeting; Tumor immunotherapy.
© 2022. The Author(s), under exclusive licence to Springer-Verlag GmbH Germany, part of Springer Nature.
Conflict of interest statement
The authors declare that there are no conflict of interest.
Figures


References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

