Understanding the Cellular Sources of the Fractional Exhaled Nitric Oxide (FeNO) and Its Role as a Biomarker of Type 2 Inflammation in Asthma
- PMID: 35547356
- PMCID: PMC9085317
- DOI: 10.1155/2022/5753524
Understanding the Cellular Sources of the Fractional Exhaled Nitric Oxide (FeNO) and Its Role as a Biomarker of Type 2 Inflammation in Asthma
Abstract
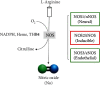
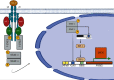
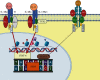
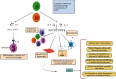
Fractional exhaled nitric oxide (FeNO) has gained great clinical importance as a biomarker of type 2 inflammation in chronic airway diseases such as asthma. FeNO originates primarily in the bronchial epithelium and is produced in large quantities by the enzyme inducible nitric oxide synthase (iNOS). It should be noted that nitric oxide (NO) produced at femtomolar to picomolar levels is fundamental for respiratory physiology. This basal production is induced in the bronchial epithelium by interferon gamma (IFNγ) via Janus kinases (JAK)/STAT-1 signaling. However, when there is an increase in the expression of type 2 inflammatory cytokines such as IL-4 and IL-13, the STAT-6 pathway is activated, leading to overexpression of iNOS and consequently to an overproduction of airway NO. Increased NO levels contributes to bronchial hyperreactivity and mucus hypersecretion, increases vascular permeability, reduces ciliary heartbeat, and promotes free radical production, airway inflammation, and tissue damage. In asthmatic patients, FeNO levels usually rise above 25 parts per billion (ppb) and its follow-up helps to define asthma phenotype and to monitor the effectiveness of corticosteroid treatment and adherence to treatment. FeNO is also very useful to identify those severe asthma patients that might benefit of personalized therapies with monoclonal antibodies. In this review, we revised the cellular and molecular mechanisms of NO production in the airway and its relevance as a biomarker of type 2 inflammation in asthma.
Copyright © 2022 Jose M. Escamilla-Gil et al.
Conflict of interest statement
The authors declare that there is no conflict of interest regarding the publication of this paper.
Figures


References
-
- American Thoracic S., European Respiratory S. ATS/ERS recommendations for standardized procedures for the online and offline measurement of exhaled lower respiratory nitric oxide and nasal nitric oxide, 2005. American Journal of Respiratory and Critical Care Medicine . 2005;171(8):912–930. doi: 10.1164/rccm.200406-710ST. - DOI - PubMed
-
- Sabatelli L., Seppala U., Sastre J., Crater G. cost-effectiveness and budget impact of routine use of fractional exhaled nitric oxide monitoring for the management of adult asthma patients in Spain. Journal of Investigational Allergology & Clinical Immunology . 2017;27(2):89–97. doi: 10.18176/jiaci.0103. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

