Polymer chimera of stapled oncolytic peptide coupled with anti-PD-L1 peptide boosts immunotherapy of colorectal cancer
- PMID: 35547769
- PMCID: PMC9065177
- DOI: 10.7150/thno.71129
Polymer chimera of stapled oncolytic peptide coupled with anti-PD-L1 peptide boosts immunotherapy of colorectal cancer
Abstract
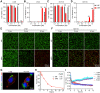
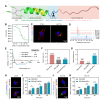
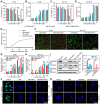
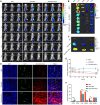
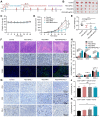
Rationale: Scarce tumor mutation burden and neoantigens create tremendous obstacles for an effective immunotherapy of colorectal cancer (CRC). Oncolytic peptides rise as a promising therapeutic approach that boosts tumor-specific immune responses by inducing antigenic substances. However, the clinical application of oncolytic peptides has been hindered because of structural instability, proteolytic degradation, and undesired toxicity when administered systemically. Methods: Based on wasp venom peptide, an optimized stapled oncolytic peptide MP9 was developed with rigid α-helix, protease-resistance, and CRC cell cytotoxicity. By incorporating four functional motifs that include D-peptidomimetic inhibitor of PD-L1, matrix metalloproteinase-2 (MMP-2) cleavable spacer, and MP9 with 4-arm PEG, a novel peptide-polymer conjugate (PEG-MP9-aPDL1) was obtained and identified as the most promising systemic delivery vehicle with PD-L1 targeting specificity and favorable pharmacokinetic properties. Results: We demonstrated that PEG-MP9-aPDL1-driven oncolysis induces a panel of immunogenic cell death (ICD)-relevant damage-associated molecular patterns (DAMPs) both in vitro and in vivo, which are key elements for immunotherapy with PD-L1 inhibitor. Further, PEG-MP9-aPDL1 exhibited prominent immunotherapeutic efficacy in a CRC mouse model characterized by tumor infiltration of CD8+ T cells and induction of cytotoxic lymphocytes (CTLs) in the spleens. Conclusion: Our findings suggest that PEG-MP9-aPDL1 is an all-in-one platform for oncolytic immunotherapy and immune checkpoint blockade (ICB).
Keywords: colorectal cancer; immunogenic cell death; oncolytic immunotherapy; peptide-polymer conjugate; stapled mastoparan peptide.
© The author(s).
Conflict of interest statement
Competing Interests: The authors have declared that no competing interest exists. The image of graphic abstract was created with Biorender.com. Other images were created with Adobe Illustrator.
Figures

References
-
- Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A. et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71:209–49. - PubMed
-
- Guo J, Yu Z, Das M, Huang L. Nano codelivery of oxaliplatin and folinic acid achieves synergistic chemo-immunotherapy with 5-fluorouracil for colorectal cancer and liver metastasis. ACS Nano. 2020;14:5075–89. - PubMed
-
- Dekker E, Tanis PJ, Vleugels JLA, Kasi PM, Wallace MB. Colorectal cancer. Lancet. 2019;394:1467–80. - PubMed
-
- Le DT, Hubbard-Lucey VM, Morse MA, Heery CR, Dwyer A, Marsilje TH. et al. A blueprint to advance colorectal Cancer Immunotherapies. Cancer Immunol Res. 2017;5:942–9. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

