Probing the Electronic Properties and Interaction Landscapes in a Series of N-(Chlorophenyl)pyridinecarboxamides
- PMID: 35547941
- PMCID: PMC9074230
- DOI: 10.1021/acs.cgd.2c00153
Probing the Electronic Properties and Interaction Landscapes in a Series of N-(Chlorophenyl)pyridinecarboxamides
Abstract
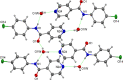
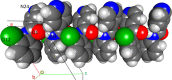
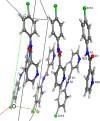
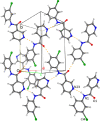
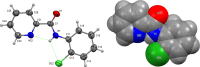
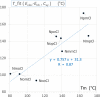
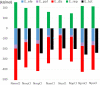
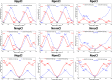
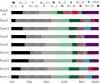
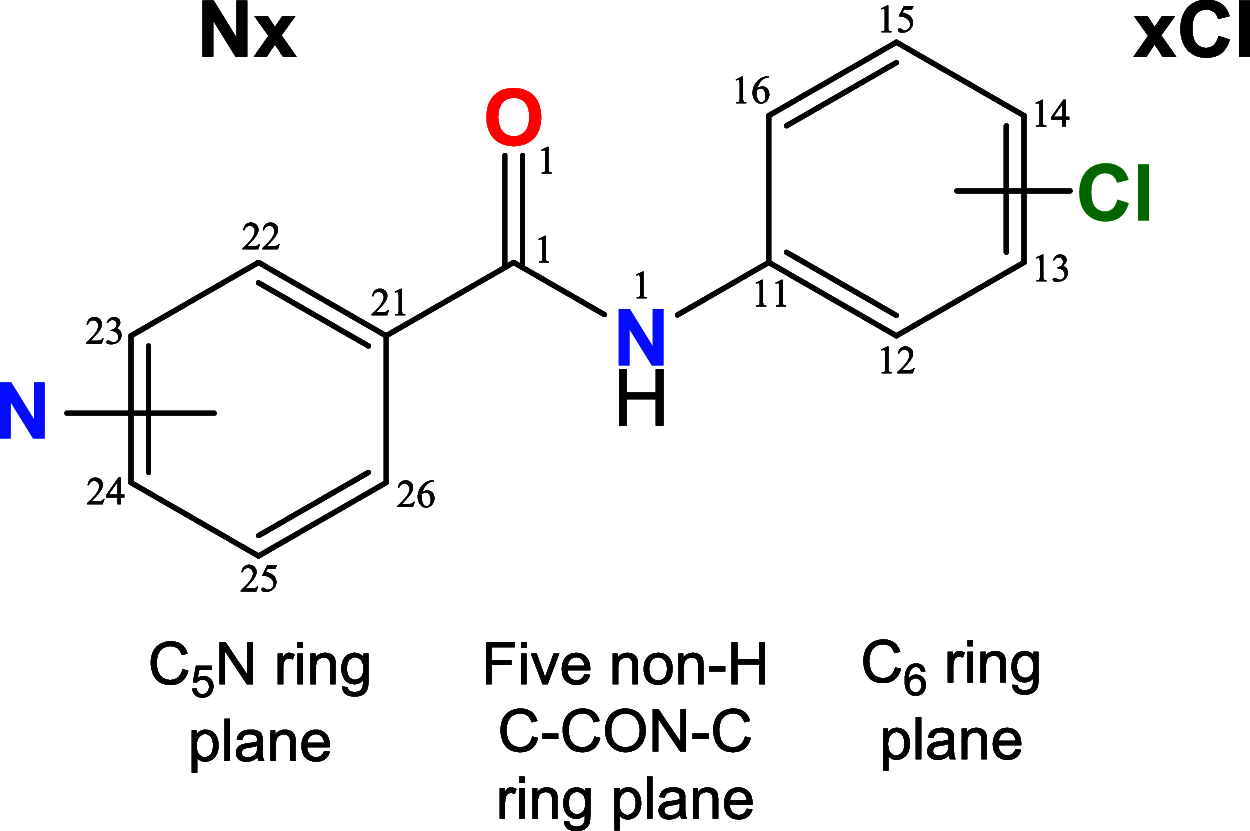
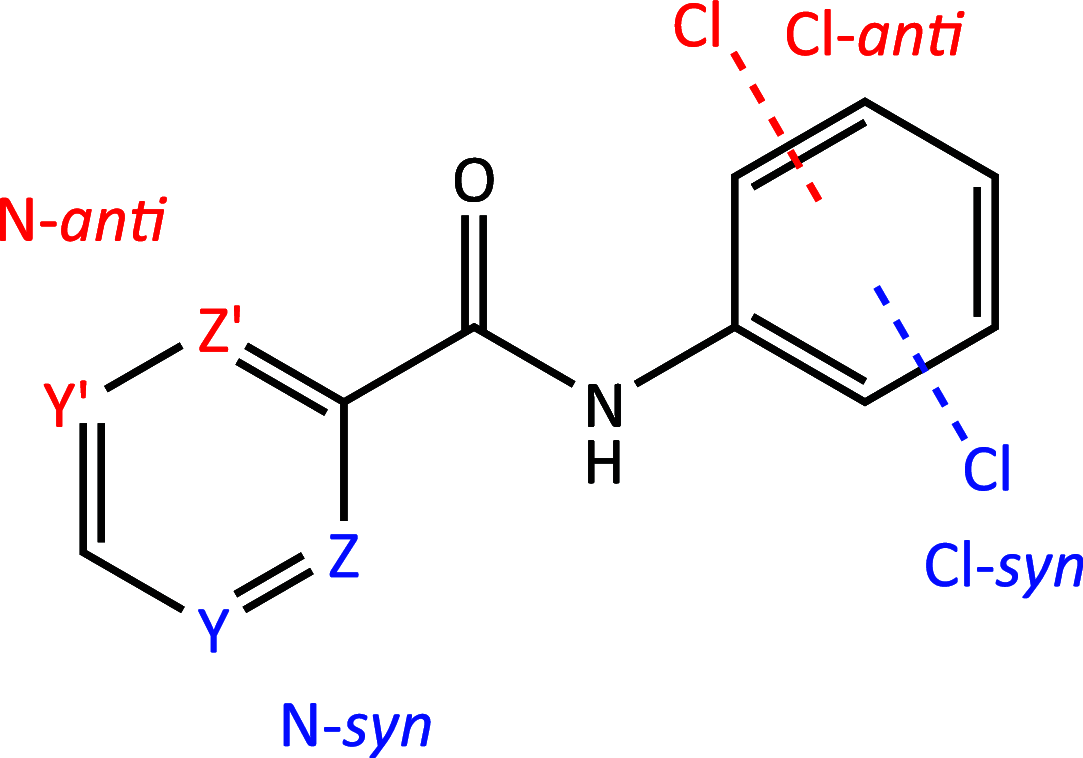
A 3 × 3 isomer grid of nine N-(chlorophenyl)pyridinecarboxamides (NxxCl) is reported with physicochemical studies and single crystal structures (Nx = pyridinoyl moiety; xCl = aminochlorobenzene ring; x = para-/meta-/ortho-), as synthesized by the reaction of the substituted p-/m-/o-pyridinecarbonyl chlorides (Nx) with p-/m-/o-aminochlorobenzenes (xCl). Several of the nine NxxCl crystal structures display structural similarities with their halogenated NxxX and methylated NxxM relatives (x = p-/m-/o-substitutions; X = F, Br; M = methyl). Indeed, five of the nine NxxCl crystal structures are isomorphous with their NxxBr analogues as the NpmCl/Br, NpoCl/Br, NmoCl/NmoBr, NopCl/Br, and NooCl/Br pairs. In the NxxCl series, the favored hydrogen bonding mode is aggregation by N-H···Npyridine interactions, though amide···amide intermolecular interactions are noted in NpoCl and NmoCl. For the NoxCl triad, intramolecular N-H···Npyridine interactions influence molecular planarity, whereas NppCl·H2O (as a monohydrate) exhibits O-H···O, N-H···O1W, and O1W-H···N interactions as the primary hydrogen bonding. Analysis of chlorine-containing compounds on the CSD is noted for comparisons. The interaction environments are probed using Hirshfeld surface analysis and contact enrichment studies. The melting temperatures (T m) depend on both the lattice energy and molecular symmetry (Carnelley's rule), and the melting points can be well predicted from a linear regression of the two variables. The relationships of the T m values with the total energy, the electrostatic component, and the strongest hydrogen bond components have been analyzed.
© 2022 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures



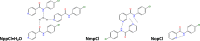
Similar articles
-
Structural systematics and conformational analyses of a 3 × 3 isomer grid of fluoro-N-(pyridyl)benzamides: physicochemical correlations, polymorphism and isomorphous relationships.Acta Crystallogr B. 2012 Apr;68(Pt 2):189-203. doi: 10.1107/S0108768112006799. Epub 2012 Mar 20. Acta Crystallogr B. 2012. PMID: 22436918
-
Supramolecular hydrogen-bonding patterns in salts of the antifolate drugs trimethoprim and pyrimethamine.Acta Crystallogr C Struct Chem. 2018 Apr 1;74(Pt 4):487-503. doi: 10.1107/S2053229618004072. Epub 2018 Mar 23. Acta Crystallogr C Struct Chem. 2018. PMID: 29620034
-
Three isomorphous 2,6-dibromopyridinium tetrabromidometallates: (C(5)H(4)Br(2)N)(2)[MBr(4)].2H(2)O (M = Cu, Cd and Hg).Acta Crystallogr C. 2009 Nov;65(Pt 11):m451-4. doi: 10.1107/S010827010903772X. Epub 2009 Oct 28. Acta Crystallogr C. 2009. PMID: 19893231
-
Synthesis, experimental and in silico studies of N-fluorenylmethoxycarbonyl-O-tert-butyl-N-methyltyrosine, coupled with CSD data: a survey of interactions in the crystal structures of Fmoc-amino acids.Acta Crystallogr C Struct Chem. 2020 Apr 1;76(Pt 4):328-345. doi: 10.1107/S2053229620003009. Epub 2020 Mar 10. Acta Crystallogr C Struct Chem. 2020. PMID: 32229714
-
Positional isomeric effect on the crystallization of chlorine-substituted N-phenyl-2-phthalimidoethanesulfonamide derivatives.Acta Crystallogr C Struct Chem. 2015 Sep;71(Pt 9):839-43. doi: 10.1107/S2053229615015223. Epub 2015 Aug 27. Acta Crystallogr C Struct Chem. 2015. PMID: 26322619
Cited by
-
Synthesis, crystal structure and Hirshfeld surface analysis of N-(4-meth-oxy-phen-yl)picolinamide.Acta Crystallogr E Crystallogr Commun. 2024 Nov 14;80(Pt 12):1293-1297. doi: 10.1107/S2056989024010843. eCollection 2024 Nov 1. Acta Crystallogr E Crystallogr Commun. 2024. PMID: 39906781 Free PMC article.
References
-
- Gribble G. W. The diversity of naturally occurring organobromine compounds. Chem. Soc. Rev. 1999, 28, 335–346. 10.1039/a900201d. - DOI
-
- Gribble G. W. A recent survey of naturally occurring organohalogen compounds. Environ. Chem. 2015, 12, 396–405. 10.1071/EN15002. - DOI
-
- Jin L. J.; Chen B. L. Natural Origins, Concentration Levels, and Formation Mechanisms of Organohalogens in the Environment. Prog. Chem. 2017, 29, 1093–1114. 10.7536/PC170563. - DOI
LinkOut - more resources
Full Text Sources
