An ''off-on-off'' sensor for sequential detection of Cu2+ and hydrogen sulfide based on a naphthalimide-rhodamine B derivative and its application in dual-channel cell imaging
- PMID: 35548160
- PMCID: PMC9086380
- DOI: 10.1039/c8ra05963b
An ''off-on-off'' sensor for sequential detection of Cu2+ and hydrogen sulfide based on a naphthalimide-rhodamine B derivative and its application in dual-channel cell imaging
Abstract
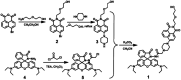
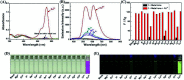
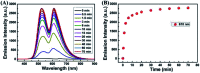
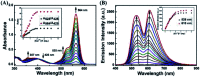
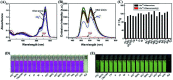
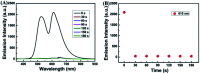
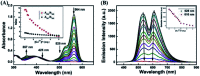
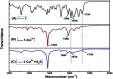
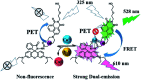
A novel colorimetric and fluorometric sensor with unique dual-channel emission to sequentially detect Cu2+ and hydrogen sulfide (H2S) was synthesized from naphthalimide-rhodamine B through the PET and FRET mechanism. The sensor showed a selective "off-on" fluorescence response with a 120-fold increase toward Cu2+, and its limits of detection were 0.26 μM and 0.17 μM for UV-vis and fluorescence measurements, respectively. In addition, 1-Cu2+ was an efficient "on-off" sensor to detect H2S with detection limits of 0.40 μM (UV-vis measurement) and 0.23 μM (fluorescence measurement), respectively. Furthermore, the sensor can also be used for biological imaging of intracellular staining in living cells. Therefore, the sensor should be highly promising for the detection of low level Cu2+ and H2S with great potential in many practical applications.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures
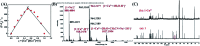

Similar articles
-
A colorimetric and ratiometric fluorescent sensor for sequentially detecting Cu2+ and arginine based on a coumarin-rhodamine B derivative and its application for bioimaging.RSC Adv. 2019 Feb 26;9(12):6643-6649. doi: 10.1039/c8ra09943j. eCollection 2019 Feb 22. RSC Adv. 2019. PMID: 35518477 Free PMC article.
-
A novel rhodamine B-based "off-on'' fluorescent sensor for selective recognition of copper (II) ions.Talanta. 2018 Jul 1;184:143-148. doi: 10.1016/j.talanta.2018.01.073. Epub 2018 Feb 2. Talanta. 2018. PMID: 29674025
-
Novel turn-on fluorescence sensor for detection and imaging of endogenous H2S induced by sodium nitroprusside.Spectrochim Acta A Mol Biomol Spectrosc. 2020 Dec 15;243:118775. doi: 10.1016/j.saa.2020.118775. Epub 2020 Aug 6. Spectrochim Acta A Mol Biomol Spectrosc. 2020. PMID: 32827912
-
FRET-based dual channel fluorescent probe for detecting endogenous/exogenous H2O2/H2S formation through multicolor images.J Photochem Photobiol B. 2019 Feb;191:99-106. doi: 10.1016/j.jphotobiol.2018.12.016. Epub 2018 Dec 26. J Photochem Photobiol B. 2019. PMID: 30597284
-
An anthraquinone-based highly selective colorimetric and fluorometric sensor for sequential detection of Cu2+ and S2- with intracellular application.J Mater Chem B. 2017 Dec 7;5(45):8957-8966. doi: 10.1039/c7tb01596h. Epub 2017 Nov 7. J Mater Chem B. 2017. PMID: 32264122
Cited by
-
Nanomolar Detection of H2S in an Aqueous Medium: Application in Endogenous and Exogenous Imaging of HeLa Cells and Zebrafish.ACS Omega. 2020 Aug 3;5(31):19896-19904. doi: 10.1021/acsomega.0c02963. eCollection 2020 Aug 11. ACS Omega. 2020. PMID: 32803086 Free PMC article.
-
Activity-Based Fluorescent Probes for Hydrogen Sulfide and Related Reactive Sulfur Species.Chem Rev. 2024 Apr 10;124(7):4124-4257. doi: 10.1021/acs.chemrev.3c00683. Epub 2024 Mar 21. Chem Rev. 2024. PMID: 38512066 Free PMC article. Review.
-
A ratiometric and colorimetric probe for detecting Hg2+ based on naphthalimide-rhodamine and its staining function in cell imaging.RSC Adv. 2019 Apr 15;9(21):11664-11669. doi: 10.1039/c9ra01459d. eCollection 2019 Apr 12. RSC Adv. 2019. PMID: 35516999 Free PMC article.
-
Review of Chemical Sensors for Hydrogen Sulfide Detection in Organisms and Living Cells.Sensors (Basel). 2023 Mar 21;23(6):3316. doi: 10.3390/s23063316. Sensors (Basel). 2023. PMID: 36992027 Free PMC article. Review.
-
A turn-on fluorescent probe with a dansyl fluorophore for hydrogen sulfide sensing.RSC Adv. 2019 Sep 3;9(47):27652-27658. doi: 10.1039/c9ra04790e. eCollection 2019 Aug 29. RSC Adv. 2019. PMID: 35529213 Free PMC article.
References
LinkOut - more resources
Full Text Sources

