Particle Safety Assessment in Additive Manufacturing: From Exposure Risks to Advanced Toxicology Testing
- PMID: 35548681
- PMCID: PMC9081788
- DOI: 10.3389/ftox.2022.836447
Particle Safety Assessment in Additive Manufacturing: From Exposure Risks to Advanced Toxicology Testing
Abstract
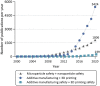
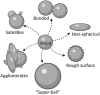
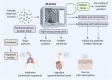
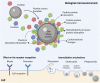
Additive manufacturing (AM) or industrial three-dimensional (3D) printing drives a new spectrum of design and production possibilities; pushing the boundaries both in the application by production of sophisticated products as well as the development of next-generation materials. AM technologies apply a diversity of feedstocks, including plastic, metallic, and ceramic particle powders with distinct size, shape, and surface chemistry. In addition, powders are often reused, which may change the particles' physicochemical properties and by that alter their toxic potential. The AM production technology commonly relies on a laser or electron beam to selectively melt or sinter particle powders. Large energy input on feedstock powders generates several byproducts, including varying amounts of virgin microparticles, nanoparticles, spatter, and volatile chemicals that are emitted in the working environment; throughout the production and processing phases. The micro and nanoscale size may enable particles to interact with and to cross biological barriers, which could, in turn, give rise to unexpected adverse outcomes, including inflammation, oxidative stress, activation of signaling pathways, genotoxicity, and carcinogenicity. Another important aspect of AM-associated risks is emission/leakage of mono- and oligomers due to polymer breakdown and high temperature transformation of chemicals from polymeric particles, both during production, use, and in vivo, including in target cells. These chemicals are potential inducers of direct toxicity, genotoxicity, and endocrine disruption. Nevertheless, understanding whether AM particle powders and their byproducts may exert adverse effects in humans is largely lacking and urges comprehensive safety assessment across the entire AM lifecycle-spanning from virgin and reused to airborne particles. Therefore, this review will detail: 1) brief overview of the AM feedstock powders, impact of reuse on particle physicochemical properties, main exposure pathways and protective measures in AM industry, 2) role of particle biological identity and key toxicological endpoints in the particle safety assessment, and 3) next-generation toxicology approaches in nanosafety for safety assessment in AM. Altogether, the proposed testing approach will enable a deeper understanding of existing and emerging particle and chemical safety challenges and provide a strategy for the development of cutting-edge methodologies for hazard identification and risk assessment in the AM industry.
Keywords: adverse outcome; endocrine disruption; genotoxicity; industrial 3D printing; inflammation; mechanism of action; particle emissions.
Copyright © 2022 Alijagic, Engwall, Särndahl, Karlsson, Hedbrant, Andersson, Karlsson, Dalemo, Scherbak, Färnlund, Larsson and Persson.
Conflict of interest statement
Author MD was employed by the Absolent AB and Author FK was employed by the AMEXCI AB. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The handling editor declared a past collaboration with one of the author AA.
Figures


References
-
- Aguilera P. A., Fernández A., Fernández R., Rumí R., Salmerón A. (2011). Bayesian Networks in Environmental Modelling. Environ. Model. Softw. 26 (12), 1376–1388. 10.1016/j.envsoft.2011.06.004 - DOI
-
- Andersson L., Bryngelsson I.-L., Hedbrant A., Persson A., Johansson A., Ericsson A., et al. (2019). Respiratory Health and Inflammatory Markers - Exposure to Respirable Dust and Quartz and Chemical Binders in Swedish Iron Foundries. PloS one 14 (11), e0224668. 10.1371/journal.pone.0224668 - DOI - PMC - PubMed
-
- Angioletti-Uberti S., Ballauff M., Dzubiella J. (2018). Competitive Adsorption of Multiple Proteins to Nanoparticles: the Vroman Effect Revisited. Mol. Phys. 116 (21-22), 3154–3163. 10.1080/00268976.2018.1467056 - DOI
-
- Ardila L. C., Garciandia F., González-Díaz J. B., Álvarez P., Echeverria A., Petite M. M., et al. (2014). Effect of IN718 Recycled Powder Reuse on Properties of Parts Manufactured by Means of Selective Laser Melting. Phys. Proced. 56, 99–107. 10.1016/j.phpro.2014.08.152 - DOI
-
- Arsalane K., Broeckaert F., Knoops B., Wiedig M., Toubeau G., Bernard A. (2000). Clara Cell Specific Protein (CC16) Expression after Acute Lung Inflammation Induced by Intratracheal Lipopolysaccharide Administration. Am. J. Respir. Crit. Care Med. 161 (5), 1624–1630. 10.1164/ajrccm.161.5.9812157 - DOI - PubMed

