Synthesis of novel cyclopeptides containing heterocyclic skeletons
- PMID: 35548835
- PMCID: PMC9086729
- DOI: 10.1039/c8ra03899f
Synthesis of novel cyclopeptides containing heterocyclic skeletons
Abstract
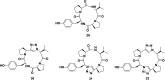
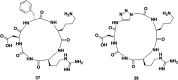
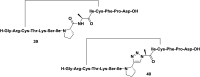
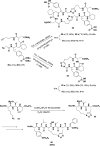
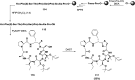
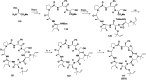
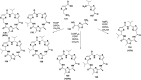
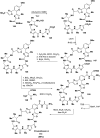
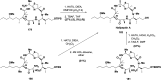
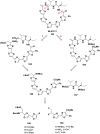
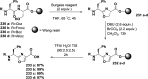
Cyclopeptides can be considered as naturally biologically active compounds. Over the last several decades, many attempts have been made to synthesize complex naturally occurring cyclopeptides, and great progress has been achieved to advance the field of total synthesis. Moreover, cyclopeptides containing heterocyclic skeletons have been recently developed into powerful reactions and approaches. This review aims to highlight recent advances in the synthesis of cyclopeptides containing heterocyclic skeletons such as triazole, oxazole, thiazole, and tetrazole.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures

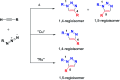
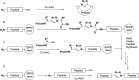
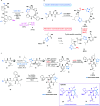

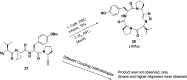





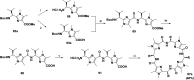

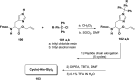
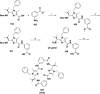
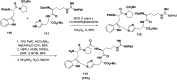
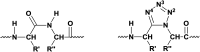





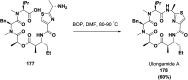











References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

