Merging Directed C-H Activations with High-Throughput Experimentation: Development of Iridium-Catalyzed C-H Aminations Applicable to Late-Stage Functionalization
- PMID: 35557751
- PMCID: PMC9088304
- DOI: 10.1021/jacsau.2c00039
Merging Directed C-H Activations with High-Throughput Experimentation: Development of Iridium-Catalyzed C-H Aminations Applicable to Late-Stage Functionalization
Abstract
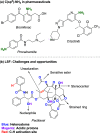
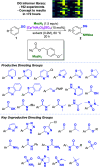
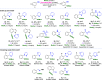
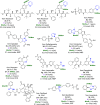
Herein, we report an iridium-catalyzed directed C-H amination methodology developed using a high-throughput experimentation (HTE)-based strategy, applicable for the needs of automated modern drug discovery. The informer library approach for investigating the accessible directing group chemical space, in combination with functional group tolerance screening and substrate scope investigations, allowed for the generation of reaction application guidelines to aid future users. Applicability to late-stage functionalization of complex drugs and natural products, in combination with multiple deprotection protocols leading to the desirable aniline matched pairs, serve to demonstrate the utility of the method for drug discovery. Finally, reaction miniaturization to a nanomolar range highlights the opportunities for more sustainable screening with decreased material consumption.
© 2022 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures



Similar articles
-
High-Throughput Enabled Iridium-Catalyzed C-H Borylation Platform for Late-Stage Functionalization.ACS Catal. 2025 Feb 12;15(4):3525-3534. doi: 10.1021/acscatal.4c07711. eCollection 2025 Feb 21. ACS Catal. 2025. PMID: 40013248 Free PMC article.
-
Late-Stage Amination of Drug-Like Benzoic Acids: Access to Anilines and Drug Conjugates through Directed Iridium-Catalyzed C-H Activation.Chemistry. 2021 Dec 23;27(72):18188-18200. doi: 10.1002/chem.202103510. Epub 2021 Nov 17. Chemistry. 2021. PMID: 34672032 Free PMC article.
-
The Evolution of Chemical High-Throughput Experimentation To Address Challenging Problems in Pharmaceutical Synthesis.Acc Chem Res. 2017 Dec 19;50(12):2976-2985. doi: 10.1021/acs.accounts.7b00428. Epub 2017 Nov 27. Acc Chem Res. 2017. PMID: 29172435
-
Decoding Directing Groups and Their Pivotal Role in C-H Activation.Chemistry. 2021 Sep 1;27(49):12453-12508. doi: 10.1002/chem.202101004. Epub 2021 Jul 13. Chemistry. 2021. PMID: 34038596 Review.
-
The emergence of the C-H functionalization strategy in medicinal chemistry and drug discovery.Chem Commun (Camb). 2021 Oct 19;57(83):10842-10866. doi: 10.1039/d1cc04083a. Chem Commun (Camb). 2021. PMID: 34596175 Review.
Cited by
-
Late-stage meta-C-H alkylation of pharmaceuticals to modulate biological properties and expedite molecular optimisation in a single step.Nat Commun. 2024 Apr 18;15(1):3349. doi: 10.1038/s41467-024-46697-8. Nat Commun. 2024. PMID: 38637496 Free PMC article.
-
Late-stage synthesis of heterobifunctional molecules for PROTAC applications via ruthenium-catalysed C‒H amidation.Nat Commun. 2023 Dec 12;14(1):8222. doi: 10.1038/s41467-023-43789-9. Nat Commun. 2023. PMID: 38086825 Free PMC article.
-
High-Throughput Enabled Iridium-Catalyzed C-H Borylation Platform for Late-Stage Functionalization.ACS Catal. 2025 Feb 12;15(4):3525-3534. doi: 10.1021/acscatal.4c07711. eCollection 2025 Feb 21. ACS Catal. 2025. PMID: 40013248 Free PMC article.
-
A Brief Introduction to Chemical Reaction Optimization.Chem Rev. 2023 Mar 22;123(6):3089-3126. doi: 10.1021/acs.chemrev.2c00798. Epub 2023 Feb 23. Chem Rev. 2023. PMID: 36820880 Free PMC article. Review.
-
Synthesis of Benzoic Acids from Electrochemically Reduced CO2 Using Heterogeneous Catalysts.ChemSusChem. 2025 Feb 1;18(3):e202401084. doi: 10.1002/cssc.202401084. Epub 2024 Nov 5. ChemSusChem. 2025. PMID: 39310956 Free PMC article.
References
LinkOut - more resources
Full Text Sources
