Applications of Friedel-Crafts reactions in total synthesis of natural products
- PMID: 35558228
- PMCID: PMC9091380
- DOI: 10.1039/c8ra07325b
Applications of Friedel-Crafts reactions in total synthesis of natural products
Abstract
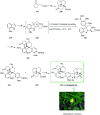
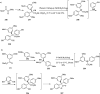
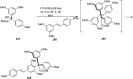
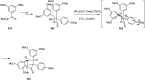
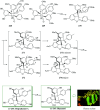
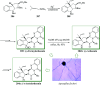
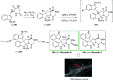
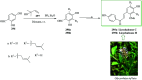
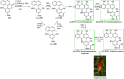
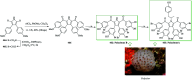
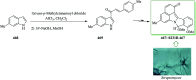
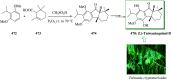
Over the years, Friedel-Crafts (FC) reactions have been acknowledged as the most useful and powerful synthetic tools for the construction of a special kind of carbon-carbon bond involving an aromatic moiety. Its stoichiometric and, more recently, its catalytic procedures have extensively been studied. This reaction in recent years has frequently been used as a key step (steps) in the total synthesis of natural products and targeted complex bioactive molecules. In this review, we try to underscore the applications of intermolecular and intramolecular FC reactions in the total syntheses of natural products and complex molecules, exhibiting diverse biological properties.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures








































Similar articles
-
New Strategies in the Efficient Total Syntheses of Polycyclic Natural Products.Acc Chem Res. 2020 Nov 17;53(11):2569-2586. doi: 10.1021/acs.accounts.0c00531. Epub 2020 Nov 2. Acc Chem Res. 2020. PMID: 33136373
-
Tandem Hock and Friedel-Crafts reactions allowing an expedient synthesis of a cyclolignan-type scaffold.Beilstein J Org Chem. 2024 Jan 25;20:162-169. doi: 10.3762/bjoc.20.15. eCollection 2024. Beilstein J Org Chem. 2024. PMID: 38292045 Free PMC article.
-
Oxidative Friedel-Crafts reaction and its application to the total syntheses of Amaryllidaceae alkaloids.J Org Chem. 2009 Mar 6;74(5):2039-45. doi: 10.1021/jo802728u. J Org Chem. 2009. PMID: 19199796
-
Aromatic C-H bond functionalization through organocatalyzed asymmetric intermolecular aza-Friedel-Crafts reaction: a recent update.Beilstein J Org Chem. 2023 Jun 28;19:956-981. doi: 10.3762/bjoc.19.72. eCollection 2023. Beilstein J Org Chem. 2023. PMID: 37404800 Free PMC article. Review.
-
The hetero-Friedel-Crafts-Bradsher Cyclizations with Formation of Ring Carbon-Heteroatom (P, S) Bonds, Leading to Organic Functional Materials.Materials (Basel). 2020 Oct 23;13(21):4751. doi: 10.3390/ma13214751. Materials (Basel). 2020. PMID: 33114241 Free PMC article. Review.
Cited by
-
Light-Driven Carbene Catalysis for the Synthesis of Aliphatic and α-Amino Ketones.Angew Chem Int Ed Engl. 2021 Aug 9;60(33):17925-17931. doi: 10.1002/anie.202105354. Epub 2021 Jul 14. Angew Chem Int Ed Engl. 2021. PMID: 34097802 Free PMC article.
-
Recent Development of Bis-Cyclometalated Chiral-at-Iridium and Rhodium Complexes for Asymmetric Catalysis.ACS Org Inorg Au. 2021 Dec 3;2(2):99-125. doi: 10.1021/acsorginorgau.1c00032. eCollection 2022 Apr 6. ACS Org Inorg Au. 2021. PMID: 36855455 Free PMC article. Review.
-
Friedel-Crafts-Type Acylation and Amidation Reactions in Strong Brønsted Acid: Taming Superelectrophiles.Molecules. 2022 Sep 14;27(18):5984. doi: 10.3390/molecules27185984. Molecules. 2022. PMID: 36144714 Free PMC article. Review.
-
Improved Process for the Continuous Acylation of 1,3-Benzodioxole.Molecules. 2024 Feb 4;29(3):726. doi: 10.3390/molecules29030726. Molecules. 2024. PMID: 38338470 Free PMC article.
-
Strain-Release-Driven Friedel-Crafts Spirocyclization of Azabicyclo[1.1.0]butanes.Angew Chem Int Ed Engl. 2022 Jan 17;61(3):e202114235. doi: 10.1002/anie.202114235. Epub 2021 Dec 3. Angew Chem Int Ed Engl. 2022. PMID: 34780681 Free PMC article.
References
-
- Friedel C. Crafts J. M. Compt. Rend. 1877;84:1392–1395.
-
- Price C. C. Org. React. 1962;3:1.
-
- Groves J. Chem. Soc. Rev. 1972;1:73–97. doi: 10.1039/CS9720100073. - DOI
-
- Eyley S. C. Ince F. Synlett. 1991;4:721–723.
-
- Heaney H. Compr. Org. Synth. 1991;2:733–752.
Publication types
LinkOut - more resources
Full Text Sources

