Intranasal Spray Characteristics for Best Drug Delivery in Patients With Chronic Rhinosinusitis
- PMID: 35560998
- PMCID: PMC9653517
- DOI: 10.1002/lary.30155
Intranasal Spray Characteristics for Best Drug Delivery in Patients With Chronic Rhinosinusitis
Abstract
Objectives: To determine parameter combinations for effective drug delivery of intranasal spray steroids to the ostiomeatal complex (OMC) and maxillary sinus (MS) in patients with chronic rhinosinusitis (CRS).
Methods: Each patient's sinonasal cavity was reconstructed from computed tomography scans. Intranasal airflow and drug particle transport were simulated using computational fluid dynamic modeling. Airflow simulations were performed at 15 Pascal inhalation pressure. Intranasal spray particles of 1-100 μm were simulated at release speeds of 1, 5, and 10 m/s from 6 release locations (Bottom, Center, Top, Lateral, Lateral-Bottom, and Lateral-Top) at a nozzle insertion depth of 15 mm. Drug delivery simulations were performed in the head tilted forward position.
Results: Maximal OMC deposition was 0.78%-12.44%, while maximal MS deposition was 0.02%-1.03% across all simulations. In general, particles between 6 and 10 μm had the best OMC (at 1 m/s particle velocity) and MS (at 10 m/s particle velocity) deposition. Particles ranging from 21 to 30 μm also had superior OMC deposition. The lateral and lateral-top spray release locations produced maximum OMC deposition, but no one release location demonstrated an increase in MS deposition.
Conclusion: This preliminary study suggests that it is challenging to determine a common set of intranasal spray parameter combinations for effective drug delivery to the OMC and MSs. Although drug particle size and spray particle velocity seem to impact particle deposition patterns, spray release location appears to vary with anatomical differences between subjects, particularly when the MS is the target location for particle deposition. Laryngoscope, 133:1036-1043, 2023.
Keywords: chronic rhinosinusitis; computational fluid dynamics; drug delivery.
© 2022 The American Laryngological, Rhinological and Otological Society, Inc.
Conflict of interest statement
This research was supported in part by the National Institute of Dental & Craniofacial Research of the National Institutes of Health under Award Number R01DE028554. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Figures

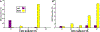



References
-
- Fokkens WJ, et al., European position paper on rhinosinusitis and nasal polyps 2020. Rhinology, 2020. 58: p. I–+. - PubMed
-
- Frank Dennis O, Z. AM, Dhandha Vishal H, McKinney Kibwei A, Fleischman Gitanjali M, Ebert Charles S Jr, Senior Brent A, Kimbell Julia S, Quantification of airflow into the maxillary sinuses before and after functional endoscopic sinus surgery. International Forum of Allergy & Rhinology, 2013. 3(10): p. 834–840. - PMC - PubMed
-
- Rudmik L, et al. Defining appropriateness criteria for endoscopic sinus surgery during management of uncomplicated adult chronic rhinosinusitis: a RAND/UCLA appropriateness study. in International Forum of Allergy & Rhinology. 2016. Wiley Online Library. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

