Beyond Kolbe and Hofer-Moest: Electrochemical Synthesis of Carboxylic Anhydrides from Carboxylic Acids
- PMID: 35561027
- PMCID: PMC9100815
- DOI: 10.1002/open.202200059
Beyond Kolbe and Hofer-Moest: Electrochemical Synthesis of Carboxylic Anhydrides from Carboxylic Acids
Abstract
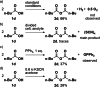
Herein we report a conceptually new non-decarboxylative electrolysis of carboxylic acids to obtain their corresponding anhydrides as highly valuable reagents in organic synthesis. All carbon atoms of the starting material are preserved in the product in an overall redox-neutral reaction. In a broad substrate scope of carboxylic acids the anhydrides are generated with high selectivity, which demonstrates the versatility of the developed method. Beneficially, no dehydrating reagents are required in comparison to conventional methods and the synthesis is based on uncritical starting materials using graphite and stainless steel as very inexpensive and eco-friendly electrode materials.
Keywords: carboxylic acids; carboxylic anhydrides; electrochemistry; electrosynthesis; sustainable chemistry.
© 2022 The Authors. Published by Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Held H., Rengstl A., Mayer D., in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, Germany, 2000, p. 3.
-
- El-Faham A., Albericio F., Chem. Rev. 2011, 111, 6557–6602. - PubMed
-
- Alizadeh A., Khodaei M. M., Nazari E., Bull. Korean Chem. Soc. 2007, 28, 1854–1856.
-
- Chen Q., Fan X.-H., Zhang L.-P., Yang L.-M., RSC Adv. 2014, 4, 53885–53890.
-
- Kocz R., Roestamadji J., Mobashery S., J. Org. Chem. 1994, 59, 2913–2914.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

