Targeted ubiquitination of sensory neuron calcium channels reduces the development of neuropathic pain
- PMID: 35561213
- PMCID: PMC9171802
- DOI: 10.1073/pnas.2118129119
Targeted ubiquitination of sensory neuron calcium channels reduces the development of neuropathic pain
Abstract
Neuropathic pain caused by lesions to somatosensory neurons due to injury or disease is a widespread public health problem that is inadequately managed by small-molecule therapeutics due to incomplete pain relief and devastating side effects. Genetically encoded molecules capable of interrupting nociception have the potential to confer long-lasting analgesia with minimal off-target effects. Here, we utilize a targeted ubiquitination approach to achieve a unique posttranslational functional knockdown of high-voltage-activated calcium channels (HVACCs) that are obligatory for neurotransmission in dorsal root ganglion (DRG) neurons. CaV-aβlator comprises a nanobody targeted to CaV channel cytosolic auxiliary β subunits fused to the catalytic HECT domain of the Nedd4-2 E3 ubiquitin ligase. Subcutaneous injection of adeno-associated virus serotype 9 encoding CaV-aβlator in the hind paw of mice resulted in the expression of the protein in a subset of DRG neurons that displayed a concomitant ablation of CaV currents and also led to an increase in the frequency of spontaneous inhibitory postsynaptic currents in the dorsal horn of the spinal cord. Mice subjected to spare nerve injury displayed a characteristic long-lasting mechanical, thermal, and cold hyperalgesia underlain by a dramatic increase in coordinated phasic firing of DRG neurons as reported by in vivo Ca2+ spike recordings. CaV-aβlator significantly dampened the integrated Ca2+ spike activity and the hyperalgesia in response to nerve injury. The results advance the principle of targeting HVACCs as a gene therapy for neuropathic pain and demonstrate the therapeutic potential of posttranslational functional knockdown of ion channels achieved by exploiting the ubiquitin-proteasome system.
Keywords: calcium channel; neuropathic pain; ubiquitin.
Conflict of interest statement
Competing interest statement: Columbia University filed a patent related to the use of engineered ubiquitin ligases as genetically encoded inhibitors for voltage-gated calcium channels.
Figures


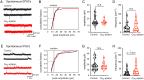
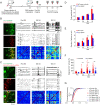
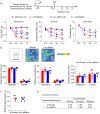
Comment in
-
The calcium channel terminator: hasta la vista pain.Trends Pharmacol Sci. 2022 Oct;43(10):801-803. doi: 10.1016/j.tips.2022.06.004. Epub 2022 Jun 23. Trends Pharmacol Sci. 2022. PMID: 35753846
References
-
- Toth C., Lander J., Wiebe S., The prevalence and impact of chronic pain with neuropathic pain symptoms in the general population. Pain Med. 10, 918–929 (2009). - PubMed
-
- van Hecke O., Austin S. K., Khan R. A., Smith B. H., Torrance N., Neuropathic pain in the general population: A systematic review of epidemiological studies. Pain 155, 654–662 (2014). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

