DNA Double-Strand Breaks as Pathogenic Lesions in Neurological Disorders
- PMID: 35563044
- PMCID: PMC9099445
- DOI: 10.3390/ijms23094653
DNA Double-Strand Breaks as Pathogenic Lesions in Neurological Disorders
Abstract
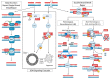
The damage and repair of DNA is a continuous process required to maintain genomic integrity. DNA double-strand breaks (DSBs) are the most lethal type of DNA damage and require timely repair by dedicated machinery. DSB repair is uniquely important to nondividing, post-mitotic cells of the central nervous system (CNS). These long-lived cells must rely on the intact genome for a lifetime while maintaining high metabolic activity. When these mechanisms fail, the loss of certain neuronal populations upset delicate neural networks required for higher cognition and disrupt vital motor functions. Mammalian cells engage with several different strategies to recognize and repair chromosomal DSBs based on the cellular context and cell cycle phase, including homologous recombination (HR)/homology-directed repair (HDR), microhomology-mediated end-joining (MMEJ), and the classic non-homologous end-joining (NHEJ). In addition to these repair pathways, a growing body of evidence has emphasized the importance of DNA damage response (DDR) signaling, and the involvement of heterogeneous nuclear ribonucleoprotein (hnRNP) family proteins in the repair of neuronal DSBs, many of which are linked to age-associated neurological disorders. In this review, we describe contemporary research characterizing the mechanistic roles of these non-canonical proteins in neuronal DSB repair, as well as their contributions to the etiopathogenesis of selected common neurological diseases.
Keywords: DNA damage response; DNA double-strand break repair; TDP-43; dementia; hnRNPs; neurodegeneration.
Conflict of interest statement
The authors declare that the study was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
-
- Yang J.L., Chen W.Y., Mukda S., Yang Y.R., Sun S.F., Chen S.D. Oxidative DNA damage is concurrently repaired by base excision repair (BER) and apyrimidinic endonuclease 1 (APE1)-initiated nonhomologous end joining (NHEJ) in cortical neurons. Neuropathol. Appl. Neurobiol. 2020;46:375–390. doi: 10.1111/nan.12584. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

