The Conformation of the N-Terminal Tails of Deinococcus grandis Dps Is Modulated by the Ionic Strength
- PMID: 35563263
- PMCID: PMC9103930
- DOI: 10.3390/ijms23094871
The Conformation of the N-Terminal Tails of Deinococcus grandis Dps Is Modulated by the Ionic Strength
Abstract
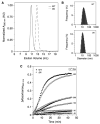
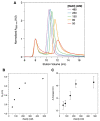
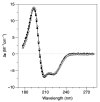
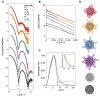
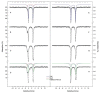
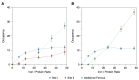
DNA-binding proteins from starved cells (Dps) are homododecameric nanocages, with N- and C-terminal tail extensions of variable length and amino acid composition. They accumulate iron in the form of a ferrihydrite mineral core and are capable of binding to and compacting DNA, forming low- and high-order condensates. This dual activity is designed to protect DNA from oxidative stress, resulting from Fenton chemistry or radiation exposure. In most Dps proteins, the DNA-binding properties stem from the N-terminal tail extensions. We explored the structural characteristics of a Dps from Deinococcus grandis that exhibits an atypically long N-terminal tail composed of 52 residues and probed the impact of the ionic strength on protein conformation using size exclusion chromatography, dynamic light scattering, synchrotron radiation circular dichroism and small-angle X-ray scattering. A novel high-spin ferrous iron-binding site was identified in the N-terminal tails, using Mössbauer spectroscopy. Our data reveals that the N-terminal tails are structurally dynamic and alter between compact and extended conformations, depending on the ionic strength of the buffer. This prompts the search for other physiologically relevant modulators of tail conformation and hints that the DNA-binding properties of Dps proteins may be affected by external factors.
Keywords: DNA-binding protein from starved cells (Dps); Deinococcus grandis; Mössbauer spectroscopy; N-terminal tail extensions; biological small-angle X-ray scattering; conformational changes; mini-ferritins.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures
References
-
- Williams S.M., Chatterji D. An Overview of Dps: Dual Acting Nanovehicles in Prokaryotes with DNA Binding and Ferroxidation Properties. In: Harris J.R., Marles-Wright J., editors. Subcellular Biochemistry. Volume 96. Springer International Publishing; Cham, Switzerland: 2021. pp. 177–216. Subcellular Biochemistry. - PubMed
-
- Guerra J.P.L., Jacinto J.P., Tavares P. Miniferritins: Small multifunctional protein cages. Coord. Chem. Rev. 2021;449:214187. doi: 10.1016/j.ccr.2021.214187. - DOI
MeSH terms
Substances
Supplementary concepts
Grants and funding
- PTDC/QUI/64248/2006/Fundação para a Ciência e Tecnologia
- UIDB/04378/2020/Fundação para a Ciência e Tecnologia
- LA/P/0140/2020/Fundação para a Ciência e Tecnologia
- PTDC/BIA-PRO/111485/2009/Fundação para a Ciência e Tecnologia
- PD/00193/2012/Fundação para a Ciência e Tecnologia
- UIDB/00068/2020/Fundação para a Ciência e Tecnologia
- PD/BD/135476/2017/Fundação para a Ciência e Tecnologia
- COVID/BD/152497/2022/Fundação para a Ciência e Tecnologia
- PD/BD/135477/2017/Fundação para a Ciência e Tecnologia
- COVID/BD/152498/2022/Fundação para a Ciência e Tecnologia
- CA15126 MOBIEU/EU HORIZON 2020
- CALIPSOplus/EU HORIZON 2020
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

