Thy-AuNP-AgNP Hybrid Systems for Colorimetric Determination of Copper (II) Ions Using UV-Vis Spectroscopy and Smartphone-Based Detection
- PMID: 35564160
- PMCID: PMC9105095
- DOI: 10.3390/nano12091449
Thy-AuNP-AgNP Hybrid Systems for Colorimetric Determination of Copper (II) Ions Using UV-Vis Spectroscopy and Smartphone-Based Detection
Abstract
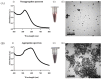
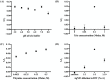
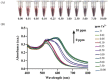
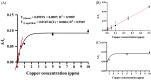
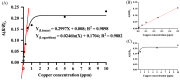
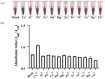
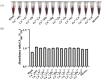
A colorimetric probe based on a hybrid sensing system of gold nanoparticles (AuNPs), silver nanoparticles (AgNPs), and thymine (Thy) was developed for easy and rapid detection of copper (II) ions (Cu2+) in solution. The underlying principle of this probe was the Cu2+-triggered aggregation of the nanoparticle components. Color change of the sensing solution (from red to purple) was clearly observed with naked eyes. The experimental parameters, including pH and concentration of tris buffer, thymine concentration and AgNP dilution ratios, were investigated and optimized. Once optimized, the limits of detection were found to be 1, 0.09 and 0.03 ppm for naked eyes, smartphone application and UV-vis spectrophotometer, respectively. Furthermore, determination of Cu2+ was accomplished within 15 min under ambient conditions. For quantitative analysis, the linearity of detection was observed through ranges of 0.09−0.5 and 0.03−0.5 ppm using smartphone application and UV-vis spectrophotometer, respectively, conforming to the World Health Organization guideline for detection of copper at concentrations < 2 ppm in water. This developed hybrid colorimetric probe exhibited preferential selectivity toward Cu2+, even when assessed in the presence of other metal ions (Al3+, Ca2+, Pb2+, Mn2+, Mg2+, Zn2+, Fe3+, Ni2+, Co2+, Hg2+ and Cd2+). The developed procedure was also successfully applied to quantification of Cu2+ in real water samples. The recovery and relative standard deviation (RSD) values from real water sample analysis were in the ranges of 70.14−103.59 and 3.21−17.63%, respectively. Our findings demonstrated a successful development and implementation of the Thy-AuNP-AgNP hybrid sensing system for rapid, simple and portable Cu2+ detection in water samples using a spectrophotometer or a smartphone-based device.
Keywords: colorimetric probe; copper (II) ion detection; gold nanoparticles; silver nanoparticles; smartphone; thymine.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Colorimetric detection of trivalent chromium in aqueous solution using tartrate-capped silver nanoparticles as probe.J Nanosci Nanotechnol. 2013 Oct;13(10):6820-5. doi: 10.1166/jnn.2013.7785. J Nanosci Nanotechnol. 2013. PMID: 24245149
-
Colorimetric detection for uranyl ions in water using vinylphosphonic acid functionalized gold nanoparticles based on smartphone.Spectrochim Acta A Mol Biomol Spectrosc. 2022 Mar 15;269:120748. doi: 10.1016/j.saa.2021.120748. Epub 2021 Dec 13. Spectrochim Acta A Mol Biomol Spectrosc. 2022. PMID: 34952439
-
A new pyrene-based Schiff-base: a selective colorimetric and fluorescent chemosensor for detection of Cu(II) and Fe(III).Spectrochim Acta A Mol Biomol Spectrosc. 2014 Sep 15;130:7-12. doi: 10.1016/j.saa.2014.03.110. Epub 2014 Apr 13. Spectrochim Acta A Mol Biomol Spectrosc. 2014. PMID: 24769379
-
Smartphone Assisted Naked Eye Detection of Mercury (II) Ion using Horseradish Peroxidase Inhibitive Assays.Protein Pept Lett. 2018;25(1):90-95. doi: 10.2174/0929866525666171214111503. Protein Pept Lett. 2018. PMID: 29237368
-
Gold Nanoparticles as Exquisite Colorimetric Transducers for Water Pollutant Detection.ACS Appl Mater Interfaces. 2023 Apr 26;15(16):19785-19806. doi: 10.1021/acsami.3c00627. Epub 2023 Apr 17. ACS Appl Mater Interfaces. 2023. PMID: 37067786 Review.
Cited by
-
Signaling strategies of silver nanoparticles in optical and electrochemical biosensors: considering their potential for the point-of-care.Mikrochim Acta. 2023 Feb 15;190(3):91. doi: 10.1007/s00604-023-05666-6. Mikrochim Acta. 2023. PMID: 36790481 Free PMC article. Review.
-
Selective detection of tricyclazole by optical technique using thiomalic acid-modified Au and Ag nanoparticle mixtures.J Food Drug Anal. 2023 Jun 15;31(2):302-314. doi: 10.38212/2224-6614.3450. J Food Drug Anal. 2023. PMID: 37335162 Free PMC article.
-
A Low-Cost Colorimetric Assay for the Analytical Determination of Copper Ions with Consumer Electronic Imaging Devices in Natural Water Samples.Molecules. 2023 Jun 17;28(12):4831. doi: 10.3390/molecules28124831. Molecules. 2023. PMID: 37375386 Free PMC article.
References
-
- Latorre M., Troncoso R., Uauy R. Clinical and Translational Perspectives on Wilson Disease. Elsevier; Amsterdam, The Netherlands: 2019. Biological Aspects of Copper; pp. 25–31. Chapter 4.
-
- Prohaska J.R. Present Knowledge in Nutrition. Wiley; Hoboken, NJ, USA: 2012. Copper; pp. 540–553.
-
- Siotto M., Squitti R. Copper imbalance in Alzheimer’s disease: Overview of the exchangeable copper component in plasma and the intriguing role albumin plays. Coord. Chem. Rev. 2018;371:86–95. doi: 10.1016/j.ccr.2018.05.020. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

