Rapid Colorimetric pH-Responsive Gold Nanocomposite Hydrogels for Sensing Applications
- PMID: 35564192
- PMCID: PMC9101415
- DOI: 10.3390/nano12091486
Rapid Colorimetric pH-Responsive Gold Nanocomposite Hydrogels for Sensing Applications
Abstract
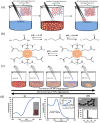
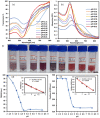
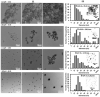
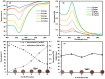
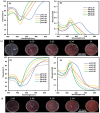
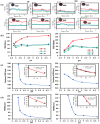
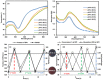
Surface functionalization of metallic nanoparticles (NPs) with external groups can be engineered to fabricate sensors that are responsive to various stimuli like temperature, pH, and numerous ions. Herein, we report the synthesis of gold nanoparticles (GNPs) functionalized with 3-mercaptopropionic acid (GNPs-MPA) and the doping of these nanoparticles into hydrogel materials using the breathing-in/breathing-out (BI-BO) method. MPA has a carboxyl group that becomes protonated and, thus, ionized at a pH below its pKa (4.32); hence, the GNPs-MPA solutions and gels were mostly pH-responsive in the range of 3-5. Optical properties were assessed through ultraviolet-visible (UV-Vis) spectroscopy, namely: transmission and absorption, and the parameters used to quantify the pH changes were the full width at half maximum (FWHM) and position of surface plasmon resonance (SPR). The solutions and gels gradually changed their colors from red to indigo with pH decrementation from 5 to 3, respectively. Furthermore, the solutions' and doped gels' highest FWHM sensitivities towards pH variations were 20 nm and 55 nm, respectively, while the SPR's position sensitivities were 18 nm and 10 nm, respectively. Also, transmission and scanning electron microscopy showed synchronized dispersion and aggregation of NPs with pH change in both solution and gel forms. The gel exhibited excellent repeatability and reversibility properties, and its response time was instantaneous, which makes its deployment as a colorimetric pH-triggered sensor practical. To the best of our knowledge, this is the first study that has incorporated GNPs into hydrogels utilizing the BI-BO method and demonstrated the pH-dependent optical and colorimetric properties of the developed nanocomposites.
Keywords: Biosensors; colorimetric sensing; nanocomposites; optical sensors; pH sensors.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
Grants and funding
LinkOut - more resources
Full Text Sources

