Latest Advances in Biomimetic Cell Membrane-Coated and Membrane-Derived Nanovectors for Biomedical Applications
- PMID: 35564251
- PMCID: PMC9104043
- DOI: 10.3390/nano12091543
Latest Advances in Biomimetic Cell Membrane-Coated and Membrane-Derived Nanovectors for Biomedical Applications
Abstract
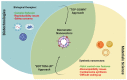
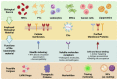
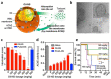
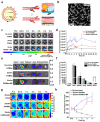
In the last decades, many nanovectors were developed for different diagnostic or therapeutic purposes. However, most nanosystems have been designed using a "bottom-up" approach, in which the basic components of the nanovector become assembled to achieve complex and specific behaviors. Despite the fine control of formulative conditions, the complexity of these systems often results cumbersome and difficult to scale-up. Recently, biomimetic materials emerged as a complementary or alternative design approach through a "top-down strategy", using cell-derived materials as building blocks to formulate innovative nanovectors. The use of cell membranes as nanoparticle coatings endows nanomaterials with the biological identity and some of the functions of the cells they are derived from. In this review, we discuss some of the latest examples of membrane coated and membrane-derived biomimetic nanomaterials and underline the common general functions offered by the biomaterials used. From these examples, we suggest a systematic classification of these biomimetic materials based on their biological sources and formulation techniques, with their respective advantages and disadvantages, and summarize the current technologies used for membranes isolation and integration on nanovectors. We also discuss some current technical limitations and hint to future direction of the improvement for biomimetics.
Keywords: biomimetic; coating; drug delivery systems; membrane; nanomedicine; nanoparticle.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

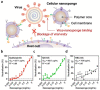

Similar articles
-
Stem cell membrane-coated abiotic nanomaterials for biomedical applications.J Control Release. 2022 Nov;351:174-197. doi: 10.1016/j.jconrel.2022.09.012. Epub 2022 Sep 22. J Control Release. 2022. PMID: 36103910 Review.
-
Engineered Cell Membrane Coating Technologies for Biomedical Applications: From Nanoscale to Macroscale.ACS Nano. 2025 Apr 1;19(12):11517-11546. doi: 10.1021/acsnano.4c16280. Epub 2025 Mar 24. ACS Nano. 2025. PMID: 40126356 Review.
-
Hybrid Membrane-Coated Biomimetic Nanoparticles (HM@BNPs): A Multifunctional Nanomaterial for Biomedical Applications.Biomacromolecules. 2021 Aug 9;22(8):3149-3167. doi: 10.1021/acs.biomac.1c00440. Epub 2021 Jul 6. Biomacromolecules. 2021. PMID: 34225451 Review.
-
Macrophage Cell Membrane-Cloaked Nanoplatforms for Biomedical Applications.Small Methods. 2022 Aug;6(8):e2200289. doi: 10.1002/smtd.202200289. Epub 2022 Jun 29. Small Methods. 2022. PMID: 35768282 Review.
-
Neuronal mitochondria-targeted delivery of curcumin by biomimetic engineered nanosystems in Alzheimer's disease mice.Acta Biomater. 2020 May;108:285-299. doi: 10.1016/j.actbio.2020.03.029. Epub 2020 Apr 3. Acta Biomater. 2020. PMID: 32251785
Cited by
-
Recent Advances in Nanodrug Delivery Systems Production, Efficacy, Safety, and Toxicity.Methods Mol Biol. 2025;2834:303-332. doi: 10.1007/978-1-0716-4003-6_15. Methods Mol Biol. 2025. PMID: 39312172 Review.
-
Cell-membrane-coated nanoparticles for the fight against pathogenic bacteria, toxins, and inflammatory cytokines associated with sepsis.Theranostics. 2023 May 21;13(10):3224-3244. doi: 10.7150/thno.81520. eCollection 2023. Theranostics. 2023. PMID: 37351162 Free PMC article. Review.
-
Nanoparticle-based approaches for treating restenosis after vascular injury.Front Pharmacol. 2024 Oct 24;15:1427651. doi: 10.3389/fphar.2024.1427651. eCollection 2024. Front Pharmacol. 2024. PMID: 39512830 Free PMC article. Review.
-
Exploring the Microfluidic Production of Biomimetic Hybrid Nanoparticles and Their Pharmaceutical Applications.Pharmaceutics. 2023 Jul 14;15(7):1953. doi: 10.3390/pharmaceutics15071953. Pharmaceutics. 2023. PMID: 37514139 Free PMC article. Review.
-
Temperature-Dependent Cytokine Neutralization Induced by Magnetoelectric Nanoparticles: An In Silico Study.Int J Mol Sci. 2024 Dec 19;25(24):13591. doi: 10.3390/ijms252413591. Int J Mol Sci. 2024. PMID: 39769353 Free PMC article.
References
-
- Baig I.K.N., Falathabe W. Nanomaterials: A review of synthesis methods, properties, recent progress, and challenges. Mater. Adv. 2021;2:1821–1871. doi: 10.1039/D0MA00807A. - DOI
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

