KRAS Mutations Impact Clinical Outcome in Metastatic Non-Small Cell Lung Cancer
- PMID: 35565194
- PMCID: PMC9103674
- DOI: 10.3390/cancers14092063
KRAS Mutations Impact Clinical Outcome in Metastatic Non-Small Cell Lung Cancer
Abstract
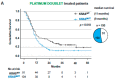
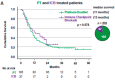
There is an urgent need to identify new predictive biomarkers for treatment response to both platinum doublet chemotherapy (PT) and immune checkpoint blockade (ICB). Here, we evaluated whether treatment outcome could be affected by KRAS mutational status in patients with metastatic (Stage IV) non-small cell lung cancer (NSCLC). All consecutive patients molecularly assessed and diagnosed between 2016−2018 with Stage IV NSCLC in the region of West Sweden were included in this multi-center retrospective study. The primary study outcome was overall survival (OS). Out of 580 Stage IV NSCLC patients, 35.5% harbored an activating mutation in the KRAS gene (KRASMUT). Compared to KRAS wild-type (KRASWT), KRASMUT was a negative factor for OS (p = 0.014). On multivariate analysis, KRASMUT persisted as a negative factor for OS (HR 1.478, 95% CI 1.207−1.709, p < 0.001). When treated with first-line platinum doublet (n = 195), KRASMUT was a negative factor for survival (p = 0.018), with median OS of 9 months vs. KRASWT at 11 months. On multivariate analysis, KRASMUT persisted as a negative factor for OS (HR 1.564, 95% CI 1.124−2.177, p = 0.008). KRASMUT patients with high PD-L1 expression (PD-L1high) had better OS than PD-L1highKRASWT patients (p = 0.036). In response to first-line ICB, KRASMUT patients had a significantly (p = 0.006) better outcome than KRASWT patients, with a median OS of 23 vs. 6 months. On multivariable Cox analysis, KRASMUT status was an independent prognostic factor for better OS (HR 0.349, 95% CI 0.148−0.822, p = 0.016). kRAS mutations are associated with better response to treatment with immune checkpoint blockade and worse response to platinum doublet chemotherapy as well as shorter general OS in Stage IV NSCLC.
Keywords: KRAS; biomarker; chemotherapy; immunotherapy; lung cancer.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
KRAS mutation predict response and outcome in advanced non-small cell lung carcinoma without driver alterations receiving PD-1 blockade immunotherapy combined with platinum-based chemotherapy: a retrospective cohort study from China.Transl Lung Cancer Res. 2022 Oct;11(10):2136-2147. doi: 10.21037/tlcr-22-655. Transl Lung Cancer Res. 2022. PMID: 36386464 Free PMC article.
-
[The efficacy and prognostic factors of immunotherapy in advanced non-small cell lung cancer patients with different driver gene mutations].Zhonghua Yi Xue Za Zhi. 2022 Apr 5;102(13):922-929. doi: 10.3760/cma.j.cn112137-20211025-02352. Zhonghua Yi Xue Za Zhi. 2022. PMID: 35385963 Chinese.
-
Oncogene-specific differences in tumor mutational burden, PD-L1 expression, and outcomes from immunotherapy in non-small cell lung cancer.J Immunother Cancer. 2021 Aug;9(8):e002891. doi: 10.1136/jitc-2021-002891. J Immunother Cancer. 2021. PMID: 34376553 Free PMC article.
-
KRAS mutation as a prognostic factor and predictive factor in advanced/metastatic non-small cell lung cancer: A systematic literature review and meta-analysis.Cancer Treat Res Commun. 2020;24:100200. doi: 10.1016/j.ctarc.2020.100200. Epub 2020 Jul 25. Cancer Treat Res Commun. 2020. PMID: 32750661
-
Immunotherapy Alone or in Combination with Chemotherapy as First-Line Treatment of Non-Small Cell Lung Cancer.Curr Treat Options Oncol. 2020 Jul 27;21(8):69. doi: 10.1007/s11864-020-00768-2. Curr Treat Options Oncol. 2020. PMID: 32720019 Review.
Cited by
-
Combinatory analysis of immune cell subsets and tumor-specific genetic variants predict clinical response to PD-1 blockade in patients with non-small cell lung cancer.Front Oncol. 2023 Feb 9;12:1073457. doi: 10.3389/fonc.2022.1073457. eCollection 2022. Front Oncol. 2023. PMID: 36844924 Free PMC article.
-
Impact of trifluoromethyl and sulfonyl groups on the biological activity of novel aryl-urea derivatives: synthesis, in-vitro, in-silico and SAR studies.Sci Rep. 2023 Oct 16;13(1):17560. doi: 10.1038/s41598-023-44753-9. Sci Rep. 2023. PMID: 37845243 Free PMC article.
-
Precision immune regulation in KRAS-mutated cancers: the final piece of the puzzle?J Exp Clin Cancer Res. 2025 Jul 3;44(1):189. doi: 10.1186/s13046-025-03444-1. J Exp Clin Cancer Res. 2025. PMID: 40611261 Free PMC article. Review.
-
Distinct metastatic organotropism shapes prognosis in lung adenocarcinoma with brain metastasis.Front Oncol. 2025 Apr 4;15:1569517. doi: 10.3389/fonc.2025.1569517. eCollection 2025. Front Oncol. 2025. PMID: 40291914 Free PMC article.
-
Comprehensive genetic variant analysis reveals combination of KRAS and LRP1B as a predictive biomarker of response to immunotherapy in patients with non-small cell lung cancer.J Exp Clin Cancer Res. 2025 Feb 27;44(1):75. doi: 10.1186/s13046-025-03342-6. J Exp Clin Cancer Res. 2025. PMID: 40011914 Free PMC article.
References
-
- World Health Organization, International Agency for Research on Cancer Globocan 2020: Lung Cancer. International Agency for Research on Cancer. [(accessed on 2 March 2021)]. Available online: http://gco.iarc.fr/today/data/factsheets/cancers/15-Lung-fact-sheet.pdf.
-
- Swedish Lung Cancer Registry. [(accessed on 1 February 2022)]. Available online: https://cancercentrum.se/samverkan/cancerdiagnoser/lunga-och-lungsack/va....
-
- El Osta B., Behera M., Kim S., Berry L.D., Sica G., Pillai R.N., Owonikoko T.K., Kris M.G., Johnson B.E., Kwiatkowski D.J., et al. Characteristics and Outcomes of Patients with Metastatic KRAS-Mutant Lung Adenocarcinomas: The Lung Cancer Mutation Consortium Experience. J. Thorac. Oncol. Off. Publ. Int. Assoc. Study Lung Cancer. 2019;14:876–889. doi: 10.1016/j.jtho.2019.01.020. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

