Hypoxia as a Modulator of Inflammation and Immune Response in Cancer
- PMID: 35565420
- PMCID: PMC9099524
- DOI: 10.3390/cancers14092291
Hypoxia as a Modulator of Inflammation and Immune Response in Cancer
Abstract
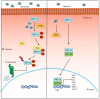
A clear association between hypoxia and cancer has heretofore been established; however, it has not been completely developed. In this sense, the understanding of the tumoral microenvironment is critical to dissect the complexity of cancer, including the reduction in oxygen distribution inside the tumoral mass, defined as tumoral hypoxia. Moreover, hypoxia not only influences the tumoral cells but also the surrounding cells, including those related to the inflammatory processes. In this review, we analyze the participation of HIF, NF-κB, and STAT signaling pathways as the main components that interconnect hypoxia and immune response and how they modulate tumoral growth. In addition, we closely examine the participation of the immune cells and how they are affected by hypoxia, the effects of the progression of cancer, and some innovative applications that take advantage of this knowledge, to suggest potential therapies. Therefore, we contribute to the understanding of the complexity of cancer to propose innovative therapeutic strategies in the future.
Keywords: HIF-1α; NF-κB; STAT; cancer; hypoxia; inflammation; tumoral microenvironment.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures





Similar articles
-
Bridging hypoxia, inflammation and estrogen receptors in thyroid cancer progression.Biomed Pharmacother. 2014 Feb;68(1):1-5. doi: 10.1016/j.biopha.2013.10.013. Epub 2013 Nov 15. Biomed Pharmacother. 2014. PMID: 24286852 Review.
-
TLR4 Agonist and Hypoxia Synergistically Promote the Formation of TLR4/NF-κB/HIF-1α Loop in Human Epithelial Ovarian Cancer.Anal Cell Pathol (Amst). 2022 Apr 14;2022:4201262. doi: 10.1155/2022/4201262. eCollection 2022. Anal Cell Pathol (Amst). 2022. PMID: 35464826 Free PMC article.
-
Hypoxia attenuates the proinflammatory response in colon cancer cells by regulating IκB.Oncotarget. 2015 Aug 21;6(24):20288-301. doi: 10.18632/oncotarget.3961. Oncotarget. 2015. PMID: 25978030 Free PMC article.
-
Crosstalk between the HIF-1 and Toll-like receptor/nuclear factor-κB pathways in the oral squamous cell carcinoma microenvironment.Oncotarget. 2016 Jun 21;7(25):37773-37789. doi: 10.18632/oncotarget.9329. Oncotarget. 2016. PMID: 27191981 Free PMC article.
-
Chronic and Cycling Hypoxia: Drivers of Cancer Chronic Inflammation through HIF-1 and NF-κB Activation: A Review of the Molecular Mechanisms.Int J Mol Sci. 2021 Oct 2;22(19):10701. doi: 10.3390/ijms221910701. Int J Mol Sci. 2021. PMID: 34639040 Free PMC article. Review.
Cited by
-
Development and Validation of a Novel Nomogram Integrated with Hypoxic and Lactate Metabolic Characteristics for Prognosis Prediction in Hepatocellular Carcinoma.J Hepatocell Carcinoma. 2024 Feb 2;11:241-255. doi: 10.2147/JHC.S446313. eCollection 2024. J Hepatocell Carcinoma. 2024. PMID: 38333220 Free PMC article.
-
Risk factor analysis and development of predictive models for osteoradionecrosis in patients with nasopharyngeal carcinoma after concurrent chemoradiotherapy.Am J Cancer Res. 2024 Oct 15;14(10):4760-4771. doi: 10.62347/RIWX7204. eCollection 2024. Am J Cancer Res. 2024. PMID: 39553231 Free PMC article.
-
Single-cell profiling reveals a conserved role for hypoxia-inducible factor signaling during human craniotomy infection.Cell Rep Med. 2024 Nov 19;5(11):101790. doi: 10.1016/j.xcrm.2024.101790. Epub 2024 Oct 18. Cell Rep Med. 2024. PMID: 39426374 Free PMC article.
-
The predictive value of pretreatment hemoglobin-to-platelet ratio on osteoradionecrosis incidence rates of locally advanced nasopharyngeal cancer patients managed with concurrent chemoradiotherapy.BMC Oral Health. 2023 Apr 20;23(1):231. doi: 10.1186/s12903-023-02937-9. BMC Oral Health. 2023. PMID: 37081475 Free PMC article.
-
A highly resolved integrated single-cell atlas of HPV-negative head and neck cancer.bioRxiv [Preprint]. 2025 Mar 4:2025.03.02.640812. doi: 10.1101/2025.03.02.640812. bioRxiv. 2025. PMID: 40093171 Free PMC article. Preprint.
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

