Parameterization and Application of the General Amber Force Field to Model Fluoro Substituted Furanose Moieties and Nucleosides
- PMID: 35565967
- PMCID: PMC9101125
- DOI: 10.3390/molecules27092616
Parameterization and Application of the General Amber Force Field to Model Fluoro Substituted Furanose Moieties and Nucleosides
Abstract
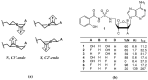
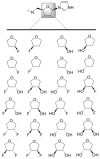
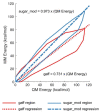
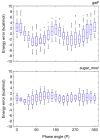
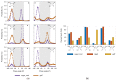
Molecular mechanics force field calculations have historically shown significant limitations in modeling the energetic and conformational interconversions of highly substituted furanose rings. This is primarily due to the gauche effect that is not easily captured using pairwise energy potentials. In this study, we present a refinement to the set of torsional parameters in the General Amber Force Field (gaff) used to calculate the potential energy of mono, di-, and gem-fluorinated nucleosides. The parameters were optimized to reproduce the pseudorotation phase angle and relative energies of a diverse set of mono- and difluoro substituted furanose ring systems using quantum mechanics umbrella sampling techniques available in the IpolQ engine in the Amber suite of programs. The parameters were developed to be internally consistent with the gaff force field and the TIP3P water model. The new set of angle and dihedral parameters and partial charges were validated by comparing the calculated phase angle probability to those obtained from experimental nuclear magnetic resonance experiments.
Keywords: Amber; NMR; fluorinated; force field; furanose; molecular mechanics; nucleoside; sugar pucker.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Improvement of RNA Simulations with Torsional Revisions of the AMBER Force Field.Methods Mol Biol. 2019;2022:55-74. doi: 10.1007/978-1-4939-9608-7_3. Methods Mol Biol. 2019. PMID: 31396899
-
A GROMOS Force Field for Furanose-Based Carbohydrates.J Chem Theory Comput. 2019 Feb 12;15(2):1168-1186. doi: 10.1021/acs.jctc.8b00838. Epub 2019 Jan 18. J Chem Theory Comput. 2019. PMID: 30609362
-
CHARMM additive all-atom force field for aldopentofuranoses, methyl-aldopentofuranosides, and fructofuranose.J Phys Chem B. 2009 Sep 17;113(37):12466-76. doi: 10.1021/jp905496e. J Phys Chem B. 2009. PMID: 19694450 Free PMC article.
-
Biomolecular force fields: where have we been, where are we now, where do we need to go and how do we get there?J Comput Aided Mol Des. 2019 Feb;33(2):133-203. doi: 10.1007/s10822-018-0111-4. Epub 2018 Nov 30. J Comput Aided Mol Des. 2019. PMID: 30506158 Review.
-
Force field development phase II: Relaxation of physics-based criteria… or inclusion of more rigorous physics into the representation of molecular energetics.J Comput Aided Mol Des. 2019 Feb;33(2):205-264. doi: 10.1007/s10822-018-0134-x. Epub 2018 Nov 30. J Comput Aided Mol Des. 2019. PMID: 30506159 Review.
References
-
- Qiu X.-L., Xu X.-H., Qing F.-L. Recent advances in the synthesis of fluorinated nucleosides. Tetrahedron. 2010;66:789–843. doi: 10.1016/j.tet.2009.11.001. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

