Ergothioneine, Ovothiol A, and Selenoneine-Histidine-Derived, Biologically Significant, Trace Global Alkaloids
- PMID: 35566030
- PMCID: PMC9103826
- DOI: 10.3390/molecules27092673
Ergothioneine, Ovothiol A, and Selenoneine-Histidine-Derived, Biologically Significant, Trace Global Alkaloids
Abstract
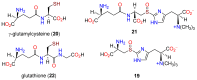
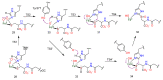
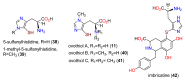
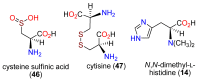
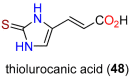
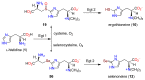
The history, chemistry, biology, and biosynthesis of the globally occurring histidine-derived alkaloids ergothioneine (10), ovothiol A (11), and selenoneine (12) are reviewed comparatively and their significance to human well-being is discussed.
Keywords: biology; biosynthesis; chemistry; distribution; ergothioneine; ovothiol A; selenoneine.
Conflict of interest statement
The authors declare no conflict of interest.
Figures











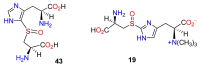

References
-
- Funayama S., Cordell G.A. Alkaloids—A Treasury of Poisons and Medicines. Academic Press; New York, NY, USA: 2015. p. 157.
-
- Santos A.P., Moreno P.R.H. Alkaloids derived from histidine: Imidazole (pilocarpine, pilosine) In: Ramawat K.G., Mérillon J.-M., editors. Handbook of Natural Products. Springer; Berlin/Heidelberg, Germany: 2013. pp. 861–882.
-
- Ferrigni N.R., Meyer B.N., McLaughlin J.L. Cactus alkaloids. LVI. Carbon-13 and proton NMR of the imidazoles, Nα,Nα-dimethylhistamine and dolichotheline. Rev. Latinoamer. Quim. 1984;14:131–133.
-
- Rosenberg H., Paul A.G. Biosynthesis of dolichotheline in Dolichothele sphaerica. Lloydia. 1971;34:372–376. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

