Lycopodium Mitigates Oxidative Stress and Inflammation in the Colonic Mucosa of Acetic Acid-Induced Colitis in Rats
- PMID: 35566122
- PMCID: PMC9102450
- DOI: 10.3390/molecules27092774
Lycopodium Mitigates Oxidative Stress and Inflammation in the Colonic Mucosa of Acetic Acid-Induced Colitis in Rats
Abstract
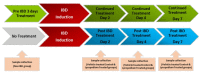
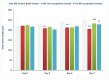
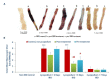
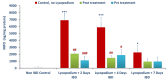
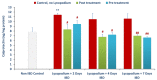
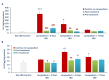
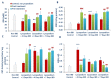
Inflammatory bowel diseases (IBDs) such as ulcerative colitis (UC) and Crohn's disease (CD) are diseases of the gastrointestinal system involving genetic and environmental factors attributed to oxidative stress and inflammation. Targeting oxidative stress and inflammation by novel dietary compounds of natural origin convincingly appears to be one of the important therapeutic strategies to keep the disease in remission. As there is no permanent cure for IBD except for chronic long-term treatment or surgery, it is therefore imperative to investigate plant-based agents that are receiving attention for their therapeutic benefits to overcome the debilitating clinical conditions of IBD. Lycopodium (LYCO), a plant of tropical and subtropical origin and known by numerous names such as ground pine, club moss, or devil's claw, has been popularly used for centuries in traditional medicine including Chinese and Indian medicines. In the present study, the effect of LYCO has been investigated in an acetic acid (AA)-induced colitis model in Wistar rats. LYCO was orally administered at the dose of 50 mg/kg/day either 3 days before or 30 min after the induction of IBD and continued for 7 days by intrarectal administration of AA. The changes in body weight and macroscopic and microscopic analysis of the colon of rats of different experimental groups were observed on days 0, 2, 4, and 7. The levels of myeloperoxidase (MPO), reduced glutathione (GSH), and malondialdehyde (MDA) were measured. AA caused a significant reduction in body weight and increased macroscopic and microscopic ulcer scores along with a significant decline in antioxidant enzymes, superoxide dismutase (SOD), and catalase and antioxidant substrate, glutathione (GSH). There was a concomitant increased formation of malondialdehyde (MDA), a marker of lipid peroxidation, and raised myeloperoxidase (MPO) activity, a marker of neutrophil activation. Treatment with LYCO significantly improved IBD-induced reduction in body weight, improved histology, inhibited MDA formation, and restored antioxidants along with reduced MPO activity. AA also caused the release of proinflammatory cytokines such as interleukin-1β (IL-1β) and interleukin-23 (IL-23). Furthermore, AA also increased the levels of calprotectin, a protein released by neutrophils under inflammatory conditions of the gastrointestinal tract. LYCO treatment significantly reduced the release of calprotectin and proinflammatory cytokines. The results demonstrate that LYCO treatment has the potential to improve disease activity by inhibiting oxidative stress, lipid peroxidation, and inflammation along with histological preservation of colonic tissues.
Keywords: IBD; colonic inflammation; gut; lycopodium; oxidative stress; plants.
Conflict of interest statement
There are no conflict of interest.
Figures


Similar articles
-
Nerolidol, a sesquiterpene, attenuates oxidative stress and inflammation in acetic acid-induced colitis in rats.Mol Cell Biochem. 2021 Sep;476(9):3497-3512. doi: 10.1007/s11010-021-04094-5. Epub 2021 May 17. Mol Cell Biochem. 2021. PMID: 33999335
-
Menthol inhibits oxidative stress and inflammation in acetic acid-induced colitis in rat colonic mucosa.Am J Transl Res. 2018 Dec 15;10(12):4210-4222. eCollection 2018. Am J Transl Res. 2018. PMID: 30662664 Free PMC article.
-
Effect of turmeric on colon histology, body weight, ulcer, IL-23, MPO and glutathione in acetic-acid-induced inflammatory bowel disease in rats.BMC Complement Altern Med. 2016 Feb 23;16:72. doi: 10.1186/s12906-016-1057-5. BMC Complement Altern Med. 2016. PMID: 26907175 Free PMC article.
-
Saffron as a Promising Therapy for Inflammatory Bowel Disease.Nutrients. 2024 Jul 20;16(14):2353. doi: 10.3390/nu16142353. Nutrients. 2024. PMID: 39064796 Free PMC article. Review.
-
Exploring the Impact of Cyanidin-3-Glucoside on Inflammatory Bowel Diseases: Investigating New Mechanisms for Emerging Interventions.Int J Mol Sci. 2023 May 28;24(11):9399. doi: 10.3390/ijms24119399. Int J Mol Sci. 2023. PMID: 37298350 Free PMC article. Review.
Cited by
-
Anti-ulcerative colitis effects of chemically characterized extracts from Calliandra haematocephala in acetic acid-induced ulcerative colitis.Front Chem. 2024 Feb 27;12:1291230. doi: 10.3389/fchem.2024.1291230. eCollection 2024. Front Chem. 2024. PMID: 38476652 Free PMC article.
-
The emerging role of oxidative stress in inflammatory bowel disease.Front Endocrinol (Lausanne). 2024 Jul 15;15:1390351. doi: 10.3389/fendo.2024.1390351. eCollection 2024. Front Endocrinol (Lausanne). 2024. PMID: 39076514 Free PMC article. Review.
-
Therapeutic Benefits of Tuna Oil by In Vitro and In Vivo Studies Using a Rat Model of Acetic Acid-Induced Ulcerative Colitis.Appl Biochem Biotechnol. 2024 Jul;196(7):3817-3843. doi: 10.1007/s12010-023-04736-y. Epub 2023 Oct 3. Appl Biochem Biotechnol. 2024. PMID: 37787891
-
Targeting inflammation in cancer therapy: from mechanistic insights to emerging therapeutic approaches.J Transl Med. 2025 May 26;23(1):588. doi: 10.1186/s12967-025-06583-3. J Transl Med. 2025. PMID: 40420174 Free PMC article. Review.
-
Anti-inflammatory effect and signaling mechanism of 8-shogaol and 10-shogaol in a dextran sodium sulfate-induced colitis mouse model.Heliyon. 2023 Jan 5;9(1):e12778. doi: 10.1016/j.heliyon.2022.e12778. eCollection 2023 Jan. Heliyon. 2023. PMID: 36647352 Free PMC article.
References
-
- Stepaniuk P., Bernstein C.N., Targownik L.E., Singh H. Characterization of Inflammatory Bowel Disease in Elderly Patients: A Review of Epidemiology, Current Practices and Outcomes of Current Management Strategies. Can. J. Gastroenterol. Hepatol. 2015;29:327–333. doi: 10.1155/2015/136960. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

