Soluble Urokinase Plasminogen Activator Receptor Levels Are Associated with Severity of Fibrosis in Patients with Primary Sclerosing Cholangitis
- PMID: 35566603
- PMCID: PMC9105770
- DOI: 10.3390/jcm11092479
Soluble Urokinase Plasminogen Activator Receptor Levels Are Associated with Severity of Fibrosis in Patients with Primary Sclerosing Cholangitis
Abstract
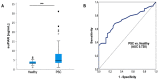
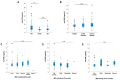
The soluble urokinase-type plasminogen activator receptor (suPAR) has evolved as a useful biomarker for different entities of chronic liver disease. However, its role in patients with primary sclerosing cholangitis (PSC) is obscure. We analyzed plasma levels of suPAR in 84 patients with PSC and compared them to 68 patients with inflammatory bowel disease (IBD) without PSC and to 40 healthy controls. Results are correlated with clinical records. suPAR concentrations were elevated in patients with PSC compared to patients with IBD only and to healthy controls (p < 0.001). Elevated suPAR levels were associated with the presence of liver cirrhosis (p < 0.001) and signs of portal hypertension (p < 0.001). suPAR revealed a high accuracy for the discrimination of the presence of liver cirrhosis comparable to previously validated noninvasive fibrosis markers (area under the curve (AUC) 0.802 (95%CI: 0.702−0.902)). Further, we demonstrated that suPAR levels may indicate the presence of acute cholangitis episodes (p < 0.001). Finally, despite the high proportion of PSC patients with IBD, presence of IBD and its disease activity did not influence circulating suPAR levels. suPAR represents a previously unrecognized biomarker for diagnosis and liver cirrhosis detection in patients with PSC. However, it does not appear to be confounded by intestinal inflammation in the context of IBD.
Keywords: biomarkers; liver cirrhosis; primary sclerosing cholangitis; soluble urokinase plasminogen activator receptor.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Circulating levels of soluble urokinase plasminogen activator receptor predict outcome after resection of biliary tract cancer.JHEP Rep. 2020 Jan 31;2(2):100080. doi: 10.1016/j.jhepr.2020.100080. eCollection 2020 Apr. JHEP Rep. 2020. PMID: 32140677 Free PMC article.
-
Circulating soluble urokinase plasminogen activator is elevated in patients with chronic liver disease, discriminates stage and aetiology of cirrhosis and predicts prognosis.Liver Int. 2012 Mar;32(3):500-9. doi: 10.1111/j.1478-3231.2011.02665.x. Epub 2011 Oct 17. Liver Int. 2012. PMID: 22098627
-
Soluble urokinase plasminogen activator receptor is compartmentally regulated in decompensated cirrhosis and indicates immune activation and short-term mortality.J Intern Med. 2013 Jul;274(1):86-100. doi: 10.1111/joim.12054. Epub 2013 Mar 17. J Intern Med. 2013. PMID: 23432143
-
Distinctive inflammatory bowel disease phenotype in primary sclerosing cholangitis.World J Gastroenterol. 2015 Feb 14;21(6):1956-71. doi: 10.3748/wjg.v21.i6.1956. World J Gastroenterol. 2015. PMID: 25684965 Free PMC article. Review.
-
The dynamic role of soluble urokinase plasminogen activator receptor (suPAR) in monitoring coagulation dysfunction during COVID-19 progression: a review.Ann Med Surg (Lond). 2024 Dec 19;87(2):635-640. doi: 10.1097/MS9.0000000000002791. eCollection 2025 Feb. Ann Med Surg (Lond). 2024. PMID: 40110334 Free PMC article. Review.
References
-
- Chapman M.H., Thorburn D., Hirschfield G.M., Webster G.G.J., Rushbrook S.M., Alexander G., Collier J., Dyson J.K., Jones D.E., Patanwala I., et al. British Society of Gastroenterology and UK-PSC guidelines for the diagnosis and management of primary sclerosing cholangitis. Gut. 2019;68:1356–1378. doi: 10.1136/gutjnl-2018-317993. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources

