Long-Term Outcomes of Implants Placed in Maxillary Sinus Floor Augmentation with Porous Fluorohydroxyapatite (Algipore® FRIOS®) in Comparison with Anorganic Bovine Bone (Bio-Oss®) and Platelet Rich Plasma (PRP): A Retrospective Study
- PMID: 35566615
- PMCID: PMC9105692
- DOI: 10.3390/jcm11092491
Long-Term Outcomes of Implants Placed in Maxillary Sinus Floor Augmentation with Porous Fluorohydroxyapatite (Algipore® FRIOS®) in Comparison with Anorganic Bovine Bone (Bio-Oss®) and Platelet Rich Plasma (PRP): A Retrospective Study
Abstract
Purpose: The objective of this retrospective study was to evaluate the long-term clinical outcomes of bone regeneration procedures using algae-derived plant hydroxyapatite (Algipore® FRIOS®) compared with demineralized anorganic bovine bone (Bio-Oss®), in combination with autologous blood-derived PRP.
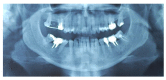
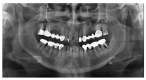
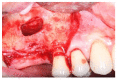
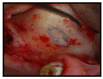
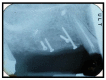
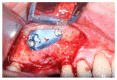
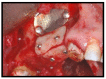
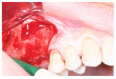
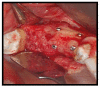
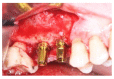
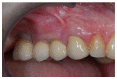
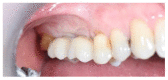
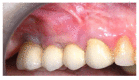
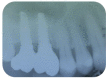
Materials and methods: Partially edentulous patients with severe atrophy of posterior maxillary treated by means of the split bone technique in a two-stage grafting procedures were observed for up to seven years after implants placement. After surgeries, the natural porous fluorohydroxyapatite (FHA) (Algipore® FRIOS®; Group, n = 29) or anorganic bovine bone (Bio-Oss® Group, n = 28) with autogenous bone in a 50:50 composite ratio with PRP, were administered in a 2.8-mm critical-size defect (CSD). Four months later, implants were placed at second-stage surgery.
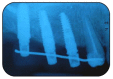
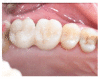
Results: A sample of fifty-seven consecutive patients who required sinus augmentation was included in the study, and 57 implants were placed. There was no drop out or loss of follow-up of any case. Clinical and radiographic examinations revealed a comparable pattern of newly formed bone in both groups after seven years of functional loading for implants placed after sinus augmentation using porous fluorohydroxyapatite and anorganic bovine bone. No significant difference in marginal bone loss was found around implants in both groups.
Conclusions: The favorable implant outcomes suggest both biomaterials are suitable for sinus grafting in severely atrophic maxillae.
Keywords: autogenous bone; biomaterials; implant; jawbone reconstruction; piezosurgery; sinus floor augmentation; vestibular region.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Sethi A., Kaus T. Maxillary ridge expansion with simultaneous implant placement: 5-year results of an ongoing clinical study. Int. J. Oral Maxillofac. Implant. 2000;15:491–499. - PubMed
-
- Anitua E., Alkhraist M.H., Piñas L., Begoña L., Orive G. Implant survival and crestal bone loss around extra-short implants supporting a fixed denture: The effect of crown height space, crown-to-implant ratio, and offset placement of the prosthesis. Int. J. Oral. Maxillofac. Implant. 2014;29:682. doi: 10.11607/jomi.3404. - DOI - PubMed
-
- Alberktsson T., Dahl E., Enbom L., Engevall S., Engquist B., Eriksson A.R., Feldmann G., Freiberg N., Glantz P.O., Kjellman O., et al. Osseointegrated oral implants. A Swedish multicenter study of 8139 consecutively inserted Nobelpharma implants. J. Periodontol. 1988;59:287. doi: 10.1902/jop.1988.59.5.287. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Research Materials

