Transcription Profile of Auxin Related Genes during Positively Gravitropic Hypocotyl Curvature of Brassica rapa
- PMID: 35567192
- PMCID: PMC9105288
- DOI: 10.3390/plants11091191
Transcription Profile of Auxin Related Genes during Positively Gravitropic Hypocotyl Curvature of Brassica rapa
Abstract
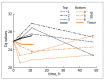
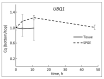
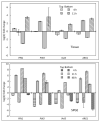
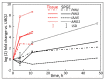
Unlike typical negative gravitropic curvature, young hypocotyls of Brassica rapa and other dicots exhibit positive gravitropism. This positive curvature occurs at the base of the hypocotyl and is followed by the typical negative gravity-induced curvature. We investigated the role of auxin in both positive and negative hypocotyl curvature by examining the transcription of PIN1, PIN3, IAA5 and ARG1 in curving tissue. We compared tissue extraction of the convex and concave flank with Solid Phase Gene Extraction (SPGE). Based on Ubiquitin1 (UBQ1) as a reference gene, the log (2) fold change of all examined genes was determined. Transcription of the examined genes varied during the graviresponse suggesting that these genes affect differential elongation. The transcription of all genes was upregulated in the lower flank and downregulated in the upper flank during the initial downward curving period. After 48 h, the transcription profile reversed, suggesting that the ensuing negative gravicurvature is controlled by the same genes as the positive gravicurvature. High-spatial resolution profiling using SPGE revealed that the transcription profile of the examined genes was spatially distinct within the curving tissue. The comparison of the hypocotyl transcription profile with the root tip indicated that the tip tissue is a suitable reference for curving hypocotyls and that root and hypocotyl curvature are controlled by the same physiological processes.
Keywords: Brassica rapa; Solid Phase Gene Extraction (SPGE); germination; gravitropism; hypocotyl; root.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




Similar articles
-
Augmentation of root gravitropism by hypocotyl curvature in Brassica rapa seedlings.Plant Sci. 2019 Aug;285:214-223. doi: 10.1016/j.plantsci.2019.05.017. Epub 2019 May 25. Plant Sci. 2019. PMID: 31203886
-
Space-time analysis of gravitropism in etiolated Arabidopsis hypocotyls using bioluminescence imaging of the IAA19 promoter fusion with a destabilized luciferase reporter.J Plant Res. 2017 Jul;130(4):765-777. doi: 10.1007/s10265-017-0932-6. Epub 2017 Apr 10. J Plant Res. 2017. PMID: 28396964 Free PMC article.
-
Evidence for a Relationship between H Excretion and Auxin in Shoot Gravitropism.Plant Physiol. 1983 May;72(1):99-104. doi: 10.1104/pp.72.1.99. Plant Physiol. 1983. PMID: 16662991 Free PMC article.
-
Auxin regulated gene expression and gravitropism in plants.ASGSB Bull. 1995 Oct;8(2):39-45. ASGSB Bull. 1995. PMID: 11538549 Review.
-
Complex physiological and molecular processes underlying root gravitropism.Plant Mol Biol. 2002 Jun-Jul;49(3-4):305-17. Plant Mol Biol. 2002. PMID: 12036256 Review.
Cited by
-
Assessing Radish Health during Space Cultivation by Gene Transcription.Plants (Basel). 2023 Sep 30;12(19):3458. doi: 10.3390/plants12193458. Plants (Basel). 2023. PMID: 37836197 Free PMC article.
-
Transcriptome and hormone metabolome reveal the mechanism of stem bending in water lily (Nymphaea tetragona) cut-flowers.Front Plant Sci. 2023 Sep 8;14:1195389. doi: 10.3389/fpls.2023.1195389. eCollection 2023. Front Plant Sci. 2023. PMID: 37746018 Free PMC article.
References
-
- Band L.R., Wells D.M., Larrieu A., Sun J.Y., Middleton A.M., French A.P., Brunoud G., Sato E.M., Wilson M.H., Peret B., et al. Root gravitropism is regulated by a transient lateral auxin gradient controlled by a tipping-point mechanism. Proc. Natl. Acad. Sci. USA. 2012;109:4668–4673. doi: 10.1073/pnas.1201498109. - DOI - PMC - PubMed
-
- Pernisova M., Prat T., Grones P., Harustiakova D., Matonohova M., Spichal L., Nodzynski T., Friml J., Hejatko J. Cytokinins influence root gravitropism via differential regulation of auxin transporter expression and localization in Arabidopsis. New Phytol. 2016;212:497–509. doi: 10.1111/nph.14049. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

