Beta interferons from the extant camelids: Unique among eutherian mammals
- PMID: 35568245
- PMCID: PMC9095258
- DOI: 10.1016/j.dci.2022.104443
Beta interferons from the extant camelids: Unique among eutherian mammals
Abstract
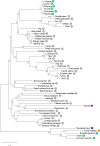
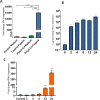
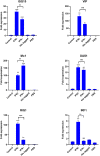
The COVID-19 pandemic is a wake-up call on the zoonotic viral spillover events and the need to be prepared for future outbreaks. Zoonotic RNA viruses like the Middle East respiratory syndrome coronavirus (MERS-CoV) are potential pathogens that could trigger the next pandemic. Dromedary camels are the only known animal source of MERS-CoV zoonotic infections, but little is known about the molecular antiviral response in this species. IFN-β and other type-I interferons provide the first line of defense against invading pathogens in the host immune response. We identified the IFNB gene of the dromedary camel and all extant members of the family Camelidae. Camelid IFN-β is unique with an even number of cysteines in the mature protein compared to other eutherian mammals with an odd number of cysteines. The viral mimetic poly(I:C) strongly induced IFN-β expression in camel kidney cells. Induction of IFN-β expression upon infection with camelpox virus was late and subdued when compared to poly(I:C) treatment. Prokaryotically expressed recombinant dromedary IFN-β induced expression of IFN-responsive genes in camel kidney cells. Further, recombinant IFN-β conferred antiviral resistance to camel kidney cells against the cytopathic effects of the camelpox virus, an endemic zoonotic pathogen. IFN-β from this unique group of mammals will offer insights into antiviral immune mechanisms and aid in the development of specific antivirals against pathogens that have the potential to be the next zoonotic pandemic.
Keywords: Antiviral; Coronavirus; Dromedary camel; Interferon; Poxvirus; Zoonosis.
Copyright © 2022 Elsevier Ltd. All rights reserved.
Figures




Similar articles
-
Novel type-I interferons from the dromedary camel: Molecular identification, prokaryotic expression and functional characterization of camelid interferon-delta.Mol Immunol. 2023 Jan;153:212-225. doi: 10.1016/j.molimm.2022.12.003. Epub 2022 Dec 21. Mol Immunol. 2023. PMID: 36563641
-
Viperin from the dromedary camel: First report of an antiviral interferon-responsive gene from camelids.Dev Comp Immunol. 2023 Oct;147:104754. doi: 10.1016/j.dci.2023.104754. Epub 2023 Jun 7. Dev Comp Immunol. 2023. PMID: 37295628
-
Camelid type I interferons: Identification and functional characterization of interferon alpha from the dromedary camel (Camelus dromedarius).Mol Immunol. 2020 Mar;119:132-143. doi: 10.1016/j.molimm.2020.01.020. Epub 2020 Jan 31. Mol Immunol. 2020. PMID: 32014632 Free PMC article.
-
Middle East Respiratory Syndrome Coronavirus (MERS-CoV) origin and animal reservoir.Virol J. 2016 Jun 3;13:87. doi: 10.1186/s12985-016-0544-0. Virol J. 2016. PMID: 27255185 Free PMC article. Review.
-
Human-Dromedary Camel Interactions and the Risk of Acquiring Zoonotic Middle East Respiratory Syndrome Coronavirus Infection.Zoonoses Public Health. 2016 Feb;63(1):1-9. doi: 10.1111/zph.12171. Epub 2014 Dec 27. Zoonoses Public Health. 2016. PMID: 25545147 Free PMC article. Review.
References
-
- Aleyas A.G., Nautiyal B., Premraj A., Rasool T.J. In: Proceedings of the 4th Conference of ISOCARD –‘Silk Road Camel: the Camelids, Main Stakes for Sustainable Development”. Special Issue of Scientific and Practical Jourmal Veterinariya #2 (42) 2015. Konuspayeva G., editor. Almaty; Kazakhstan: 2015. Development of camel renal cell line using different immortalizing agents; pp. 95–96.
-
- Aleyas A.G., Premraj A., Nautiyal B., Rasool T.J. In: Proceedings of 4th Conference of ISOCARD “Silk Road Camel: the Camelids, Main Stakes for Sustainable Development.” Special Issue of Scientific and Practical Jourmal Veterinariya #2 (42) 2015. Konuspayeva G., editor. Almaty; Kazakhstan: 2015. Endemic camel pox variants of Al Ain region; pp. 228–230.
-
- Camp J.V., Weidinger P., Ramaswamy S., Kannan D.O., Osman B.M., Kolodziejek J., Karuvantevida N., Tayoun A.A., Loney T., Nowotny N. Association of dromedary camels and camel ticks with reassortant Crimean-Congo Hemorrhagic Fever Virus, United Arab Emirates. Emerg. Infect. Dis. 2021;27:2471–2474. doi: 10.3201/eid2709.210299. - DOI - PMC - PubMed
-
- DeWitte-Orr S.J., Collins S.E., Bauer C.M.T., Bowdish D.M., Mossman K.L. An accessory to the “Trinity”: SR-As are essential pathogen sensors of extracellular dsRNA, mediating entry and leading to subsequent type I IFN responses. PLoS Pathog. 2010;6 doi: 10.1371/journal.ppat.1000829. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

