An injectable photo-cross-linking silk hydrogel system augments diabetic wound healing in orthopaedic surgery through spatiotemporal immunomodulation
- PMID: 35568914
- PMCID: PMC9107711
- DOI: 10.1186/s12951-022-01414-9
An injectable photo-cross-linking silk hydrogel system augments diabetic wound healing in orthopaedic surgery through spatiotemporal immunomodulation
Abstract
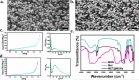
Background: The complicated hyperglycaemic and chronic inflammation of diabetic wounds in orthopaedic surgery leads to dysregulated immune cell function and potential infection risk. Immune interventions in diabetic wounds face a possible contradiction between simultaneous establishment of the pro-inflammatory microenvironment in response to potential bacterial invasion and the anti-inflammatory microenvironment required for tissue repair. To study this contradiction and accelerate diabetic-wound healing, we developed a photocurable methacryloxylated silk fibroin hydrogel (Sil-MA) system, co-encapsulated with metformin-loaded mesoporous silica microspheres (MET@MSNs) and silver nanoparticles (Ag NPs).
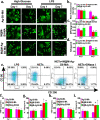
Results: The hydrogel system (M@M-Ag-Sil-MA) enhanced diabetic-wound healing via spatiotemporal immunomodulation. Sil-MA imparts a hydrogel system with rapid in situ Ultra-Violet-photocurable capability and allows preliminary controlled release of Ag NPs, which can inhibit bacterial aggregation and create a stable, sterile microenvironment. The results confirmed the involvement of Met in the immunomodulatory effects following spatiotemporal dual-controlled release via the mesoporous silica and Sil-MA. Hysteresis-released from Met shifts the M1 phenotype of macrophages in regions of diabetic trauma to an anti-inflammatory M2 phenotype. Simultaneously, the M@M-Ag-Sil-MA system inhibited the formation of neutrophil extracellular traps (NETs) and decreased the release of neutrophil elastase, myeloperoxidase, and NETs-induced pro-inflammatory factors. As a result of modulating the immune microenvironmental, the M@M-Ag-Sil-MA system promoted fibroblast migration and endothelial cell angiogenesis in vivo, with verification of enhanced diabetic-wound healing accompanied with the spatiotemporal immunoregulation of macrophages and NETs in a diabetic mouse model.
Conclusions: Our findings demonstrated that the M@M-Ag-Sil-MA hydrogel system resolved the immune contradiction in diabetic wounds through spatiotemporal immunomodulation of macrophages and NETs, suggesting its potential as a promising engineered nano-dressing for the treatment of diabetic wounds in orthopaedic surgery.
Keywords: Macrophage polarisation; Metformin; Neutrophil extracellular traps; Orthopaedic wound; Spatiotemporal immunomodulation.
© 2022. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures







Similar articles
-
A composite hydrogel containing resveratrol-laden nanoparticles and platelet-derived extracellular vesicles promotes wound healing in diabetic mice.Acta Biomater. 2022 Dec;154:212-230. doi: 10.1016/j.actbio.2022.10.038. Epub 2022 Oct 27. Acta Biomater. 2022. PMID: 36309190
-
Ag nanocomposite hydrogels with immune and regenerative microenvironment regulation promote scarless healing of infected wounds.J Nanobiotechnology. 2023 Nov 19;21(1):435. doi: 10.1186/s12951-023-02209-2. J Nanobiotechnology. 2023. PMID: 37981675 Free PMC article.
-
An Injectable Ibuprofen Sustained-Release Composite Hydrogel System Effectively Accelerates Diabetic Wound Healing via Anti-Inflammatory Effects and Angiogenesis.Int J Nanomedicine. 2025 Apr 11;20:4535-4550. doi: 10.2147/IJN.S504924. eCollection 2025. Int J Nanomedicine. 2025. PMID: 40236520 Free PMC article.
-
Immunomodulation in diabetic wounds healing: The intersection of macrophage reprogramming and immunotherapeutic hydrogels.J Tissue Eng. 2024 Jul 27;15:20417314241265202. doi: 10.1177/20417314241265202. eCollection 2024 Jan-Dec. J Tissue Eng. 2024. PMID: 39071896 Free PMC article. Review.
-
Hydrogel-based immunoregulation of macrophages for tissue repair and regeneration.Int J Biol Macromol. 2024 May;268(Pt 1):131643. doi: 10.1016/j.ijbiomac.2024.131643. Epub 2024 Apr 21. Int J Biol Macromol. 2024. PMID: 38643918 Review.
Cited by
-
Immunomodulating Hydrogels as Stealth Platform for Drug Delivery Applications.Pharmaceutics. 2022 Oct 21;14(10):2244. doi: 10.3390/pharmaceutics14102244. Pharmaceutics. 2022. PMID: 36297679 Free PMC article. Review.
-
Immunomodulatory Nanosystems: Advanced Delivery Tools for Treating Chronic Wounds.Research (Wash D C). 2023 Jul 14;6:0198. doi: 10.34133/research.0198. eCollection 2023. Research (Wash D C). 2023. PMID: 37456931 Free PMC article. Review.
-
In situ light-activated materials for skin wound healing and repair: A narrative review.Bioeng Transl Med. 2024 Jan 16;9(3):e10637. doi: 10.1002/btm2.10637. eCollection 2024 May. Bioeng Transl Med. 2024. PMID: 38818119 Free PMC article. Review.
-
Neutrophil extracellular traps in tumor metabolism and microenvironment.Biomark Res. 2025 Jan 23;13(1):12. doi: 10.1186/s40364-025-00731-z. Biomark Res. 2025. PMID: 39849606 Free PMC article. Review.
-
Advancements and Applications of Injectable Hydrogel Composites in Biomedical Research and Therapy.Gels. 2023 Jun 30;9(7):533. doi: 10.3390/gels9070533. Gels. 2023. PMID: 37504412 Free PMC article. Review.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

