Protein Kinase Inhibitor-Mediated Immunoprophylactic and Immunotherapeutic Control of Colon Cancer
- PMID: 35572581
- PMCID: PMC9097540
- DOI: 10.3389/fimmu.2022.875764
Protein Kinase Inhibitor-Mediated Immunoprophylactic and Immunotherapeutic Control of Colon Cancer
Abstract
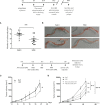
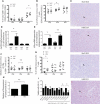
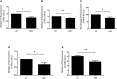
Immunotherapy has allowed major advances in oncology in the past years, in particular with the development of immune checkpoint inhibitors, but the clinical benefits are still limited, particularly in colorectal cancer (CRC). Our scientific approach is based on the search for innovative immunotherapy with a final goal that aims to induce an effective antitumor immune response in CRC. Here, we focused on a multikinase inhibitor, H89. We carried out in vivo experiments based on syngeneic mouse models of colon cancer in BALB/c mice and chemically colon tumorigenesis. Flow cytometry, RNAseq, RT-qPCR, antibody-specific immune cell depletion, and Western blot were used to identify the immune cell type involved in the preventive and antitumor activity of H89. We demonstrated that H89 delays colon oncogenesis and prevents tumor growth. This latter effect seems to involve NK cells. H89 also inhibits colon tumor growth in a T-cell-dependent manner. Analysis of the immune landscape in the tumor microenvironment showed an increase of CD4+ Th1 cells and CD8+ cytotoxic T cells but a decrease of CD4+ Treg cell infiltration. Mechanistically, we showed that H89 could promote naïve CD4+ T-cell differentiation into Th1, a decrease in Treg differentiation, and an increase in CD8+ T-cell activation and cytotoxicity ex vivo. Furthermore, H89 induced overexpression of genes involved in antitumor immune response, such as IL-15RA, which depletion counteracts the antitumor effect of H89. We also found that H89 regulated Akt/PP2A pathway axis, involved in TCR and IL-15 signaling transduction. Our findings identify the H89 as a potential strategy for immune system activation leading to the prevention and treatment of CRC.
Keywords: H89; colorectal cancer; immunoprophylaxis; immunotherapy; kinase inhibitor.
Copyright © 2022 Ghione, Racoeur, Mabrouk, Shan, Groetz, Ballot, Truntzer, Chouchane, Végran, Paul, Plenchette and Bettaieb.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

