Endogenous Retroviruses Augment Amphibian (Xenopus laevis) Tadpole Antiviral Protection
- PMID: 35575553
- PMCID: PMC9175618
- DOI: 10.1128/jvi.00634-22
Endogenous Retroviruses Augment Amphibian (Xenopus laevis) Tadpole Antiviral Protection
Abstract
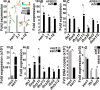
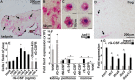
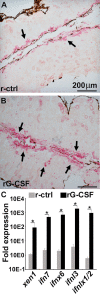
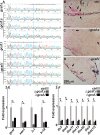
The global amphibian declines are compounded by infections with members of the Ranavirus genus such as Frog Virus 3 (FV3). Premetamorphic anuran amphibians are believed to be significantly more susceptible to FV3 while this pathogen targets the kidneys of both pre- and postmetamorphic animals. Paradoxically, FV3-challenged Xenopus laevis tadpoles exhibit lower kidney viral loads than adult frogs. Presently, we demonstrate that X. laevis tadpoles are intrinsically more resistant to FV3 kidney infections than cohort-matched metamorphic and postmetamorphic froglets and that this resistance appears to be epigenetically conferred by endogenous retroviruses (ERVs). Using a X. laevis kidney-derived cell line, we show that enhancing ERV gene expression activates cellular double-stranded RNA-sensing pathways, resulting in elevated mRNA levels of antiviral interferon (IFN) cytokines and thus greater anti-FV3 protection. Finally, our results indicate that large esterase-positive myeloid-lineage cells, rather than renal cells, are responsible for the elevated ERV/IFN axis seen in the tadpole kidneys. This conclusion is supported by our observation that CRISPR-Cas9 ablation of colony-stimulating factor-3 results in abolished homing of these myeloid cells to tadpole kidneys, concurrent with significantly abolished tadpole kidney expression of both ERVs and IFNs. We believe that the manuscript marks an important step forward in understanding the mechanisms controlling amphibian antiviral defenses and thus susceptibility and resistance to pathogens like FV3. IMPORTANCE Global amphibian biodiversity is being challenged by pathogens like the Frog Virus 3 (FV3) ranavirus, underlining the need to gain a greater understanding of amphibian antiviral defenses. While it was previously believed that anuran (frog/toad) amphibian tadpoles are more susceptible to FV3, we demonstrated that tadpoles are in fact more resistant to this virus than metamorphic and postmetamorphic froglets. We showed that this resistance is conferred by large myeloid cells within the tadpole kidneys (central FV3 target), which possess an elevated expression of endogenous retroviruses (ERVs). In turn, these ERVs activate cellular double-stranded RNA-sensing pathways, resulting in a greater expression of antiviral interferon cytokines, thereby offering the observed anti-FV3 protection.
Keywords: amphibian immunity; antiviral responses; endogenous retroviruses; interferon response.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Prominent amphibian (Xenopus laevis) tadpole type III interferon response to the frog virus 3 ranavirus.J Virol. 2015 May;89(9):5072-82. doi: 10.1128/JVI.00051-15. Epub 2015 Feb 25. J Virol. 2015. PMID: 25717104 Free PMC article.
-
Amphibian (Xenopus laevis) Tadpoles and Adult Frogs Differ in Their Antiviral Responses to Intestinal Frog Virus 3 Infections.Front Immunol. 2021 Aug 20;12:737403. doi: 10.3389/fimmu.2021.737403. eCollection 2021. Front Immunol. 2021. PMID: 34489981 Free PMC article.
-
The amphibian (Xenopus laevis) type I interferon response to frog virus 3: new insight into ranavirus pathogenicity.J Virol. 2014 May;88(10):5766-77. doi: 10.1128/JVI.00223-14. Epub 2014 Mar 12. J Virol. 2014. PMID: 24623410 Free PMC article.
-
Immune evasion strategies of ranaviruses and innate immune responses to these emerging pathogens.Viruses. 2012 Jul;4(7):1075-92. doi: 10.3390/v4071075. Epub 2012 Jun 28. Viruses. 2012. PMID: 22852041 Free PMC article. Review.
-
Antiviral immunity in amphibians.Viruses. 2011 Nov;3(11):2065-2086. doi: 10.3390/v3112065. Epub 2011 Oct 31. Viruses. 2011. PMID: 22163335 Free PMC article. Review.
Cited by
-
Xenopus laevis (Daudin, 1802) as a Model Organism for Bioscience: A Historic Review and Perspective.Biology (Basel). 2023 Jun 20;12(6):890. doi: 10.3390/biology12060890. Biology (Basel). 2023. PMID: 37372174 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

