Association of Brain Atrophy With Disease Progression Independent of Relapse Activity in Patients With Relapsing Multiple Sclerosis
- PMID: 35575778
- PMCID: PMC9112138
- DOI: 10.1001/jamaneurol.2022.1025
Association of Brain Atrophy With Disease Progression Independent of Relapse Activity in Patients With Relapsing Multiple Sclerosis
Abstract
Importance: The mechanisms driving neurodegeneration and brain atrophy in relapsing multiple sclerosis (RMS) are not completely understood.
Objective: To determine whether disability progression independent of relapse activity (PIRA) in patients with RMS is associated with accelerated brain tissue loss.
Design, setting, and participants: In this observational, longitudinal cohort study with median (IQR) follow-up of 3.2 years (2.0-4.9), data were acquired from January 2012 to September 2019 in a consortium of tertiary university and nonuniversity referral hospitals. Patients were included if they had regular clinical follow-up and at least 2 brain magnetic resonance imaging (MRI) scans suitable for volumetric analysis. Data were analyzed between January 2020 and March 2021.
Exposures: According to the clinical evolution during the entire observation, patients were classified as those presenting (1) relapse activity only, (2) PIRA episodes only, (3) mixed activity, or (4) clinical stability.
Main outcomes and measures: Mean difference in annual percentage change (MD-APC) in brain volume/cortical thickness between groups, calculated after propensity score matching. Brain atrophy rates, and their association with the variables of interest, were explored with linear mixed-effect models.
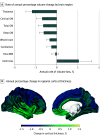
Results: Included were 1904 brain MRI scans from 516 patients with RMS (67.4% female; mean [SD] age, 41.4 [11.1] years; median [IQR] Expanded Disability Status Scale score, 2.0 [1.5-3.0]). Scans with insufficient quality were excluded (n = 19). Radiological inflammatory activity was associated with increased atrophy rates in several brain compartments, while an increased annualized relapse rate was linked to accelerated deep gray matter (GM) volume loss. When compared with clinically stable patients, patients with PIRA had an increased rate of brain volume loss (MD-APC, -0.36; 95% CI, -0.60 to -0.12; P = .02), mainly driven by GM loss in the cerebral cortex. Patients who were relapsing presented increased whole brain atrophy (MD-APC, -0.18; 95% CI, -0.34 to -0.02; P = .04) with respect to clinically stable patients, with accelerated GM loss in both cerebral cortex and deep GM. No differences in brain atrophy rates were measured between patients with PIRA and those presenting relapse activity.
Conclusions and relevance: Our study shows that patients with RMS and PIRA exhibit accelerated brain atrophy, especially in the cerebral cortex. These results point to the need to recognize the insidious manifestations of PIRA in clinical practice and to further evaluate treatment strategies for patients with PIRA in clinical trials.
Conflict of interest statement
Figures


References
-
- Kappos L, Wolinsky JS, Giovannoni G, et al. Contribution of relapse-independent progression vs relapse-associated worsening to overall confirmed disability accumulation in typical relapsing multiple sclerosis in a pooled analysis of 2 randomized clinical trials. JAMA Neurol. 2020;77(9):1132-1140. doi: 10.1001/jamaneurol.2020.1568 - DOI - PMC - PubMed
-
- Kappos L, Butzkueven H, Wiendl H, et al. ; Tysabri® Observational Program (TOP) Investigators . Greater sensitivity to multiple sclerosis disability worsening and progression events using a roving versus a fixed reference value in a prospective cohort study. Mult Scler. 2018;24(7):963-973. doi: 10.1177/1352458517709619 - DOI - PMC - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

