Mifepristone and Misoprostol for Undesired Pregnancy of Unknown Location
- PMID: 35576336
- PMCID: PMC9015019
- DOI: 10.1097/AOG.0000000000004756
Mifepristone and Misoprostol for Undesired Pregnancy of Unknown Location
Abstract
Objective: To compare immediate initiation with delayed initiation of medication abortion among patients with an undesired pregnancy of unknown location.
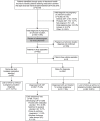
Methods: This retrospective cohort study used electronic medical record data from the Planned Parenthood League of Massachusetts (2014-2019) for patients who requested medication abortion with a last menstrual period (LMP) of 42 days or less and pregnancy of unknown location (no gestational sac) on initial ultrasonogram. Clinicians could initiate medication abortion with mifepristone followed by misoprostol while simultaneously excluding ectopic pregnancy with serial serum human chorionic gonadotropin (hCG) testing (same-day-start group) or establish a diagnosis with serial hCG tests and repeat ultrasonogram before initiating treatment (delay-for-diagnosis group). We compared primary safety outcomes (time to diagnosis of pregnancy location [rule out ectopic], emergency department visits, adverse events, and nonadherence with follow-up) between groups. We also reported secondary efficacy outcomes: time to complete abortion, successful medication abortion (no uterine aspiration), and ongoing pregnancy.
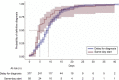
Results: Of 5,619 medication abortion visits for patients with an LMP of 42 days or less, 452 patients had pregnancy of unknown location (8.0%). Three patients underwent immediate uterine aspiration, 55 had same-day start, and 394 had delay for diagnosis. Thirty-one patients (7.9%), all in the delay-for-diagnosis group, were treated for ectopic pregnancy, including four that were ruptured. Among patients with no major ectopic pregnancy risk factors (n=432), same-day start had shorter time to diagnosis (median 5.0 days vs 9.0 days; P=.005), with no significant difference in emergency department visits (adjusted odds ratio [aOR] 0.90, 95% CI 0.43-1.88) or nonadherence with follow-up (aOR 0.92, 95% CI 0.39-2.15). Among patients who proceeded with abortion (n=270), same-day start had shorter time to complete abortion (median 5.0 days vs 19.0 days; P<.001). Of those who had medication abortion with known outcome (n=170), the rate of successful medication abortion was lower (85.4% vs 96.7%; P=.013) and the rate of ongoing pregnancy was higher (10.4% vs 2.5%; P=.041) among patients in the same-day-start group.
Conclusion: In patients with undesired pregnancy of unknown location, immediate initiation of medication abortion is associated with more rapid exclusion of ectopic pregnancy and pregnancy termination but lower abortion efficacy.
Copyright © 2022 The Author(s). Published by Wolters Kluwer Health, Inc.
Conflict of interest statement
Financial Disclosure Alisa B. Goldberg was supported by the Society of Family Planning Research Fund [SFPRF12-MA7], which was awarded to the Planned Parenthood League of Massachusetts. She received royalties from UpToDate and was a consultant for Sanofi/Genzyme from 2018 to 2019. Danielle Roncari reports receiving payment from Organon as a Nexplanon trainer. The following authors are or were employed by Planned Parenthood League of Massachusetts during the study: Jennifer Fortin, Rebecca Hofer, and Alex Cottrill. The other authors did not report any potential conflicts of interest.
Figures

Comment in
-
Mifepristone and Misoprostol for Undesired Pregnancy of Unknown Location.Obstet Gynecol. 2022 Sep 1;140(3):519. doi: 10.1097/AOG.0000000000004906. Obstet Gynecol. 2022. PMID: 36356239 No abstract available.

