Longitudinal analysis of blood DNA methylation identifies mechanisms of response to tumor necrosis factor inhibitor therapy in rheumatoid arthritis
- PMID: 35576644
- PMCID: PMC9118662
- DOI: 10.1016/j.ebiom.2022.104053
Longitudinal analysis of blood DNA methylation identifies mechanisms of response to tumor necrosis factor inhibitor therapy in rheumatoid arthritis
Abstract
Background: Rheumatoid arthritis (RA) is a chronic, immune-mediated inflammatory disease of the joints that has been associated with variation in the peripheral blood methylome. In this study, we aim to identify epigenetic variation that is associated with the response to tumor necrosis factor inhibitor (TNFi) therapy.
Methods: Peripheral blood genome-wide DNA methylation profiles were analyzed in a discovery cohort of 62 RA patients at baseline and at week 12 of TNFi therapy. DNA methylation of individual CpG sites and enrichment of biological pathways were evaluated for their association with drug response. Using a novel cell deconvolution approach, altered DNA methylation associated with TNFi response was also tested in the six main immune cell types in blood. Validation of the results was performed in an independent longitudinal cohort of 60 RA patients.
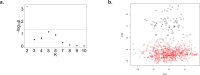
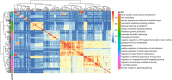
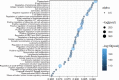
Findings: Treatment with TNFi was associated with significant longitudinal peripheral blood methylation changes in biological pathways related to RA (FDR<0.05). 139 biological functions were modified by therapy, with methylation levels changing systematically towards a signature similar to that of healthy controls. Differences in the methylation profile of T cell activation and differentiation, GTPase-mediated signaling, and actin filament organization pathways were associated with the clinical response to therapy. Cell type deconvolution analysis identified CpG sites in CD4+T, NK, neutrophils and monocytes that were significantly associated with the response to TNFi.
Interpretation: Our results show that treatment with TNFi restores homeostatic blood methylation in RA. The clinical response to TNFi is associated to methylation variation in specific biological pathways, and it involves cells from both the innate and adaptive immune systems.
Funding: The Instituto de Salud Carlos III.
Keywords: DNA methylation; Epigenetics; Rheumatoid arthritis; TNF inhibitors; Treatment response.
Copyright © 2022 The Authors. Published by Elsevier B.V. All rights reserved.
Conflict of interest statement
SM and RMM are co-founders of IMIDomics, Inc. AJ is Chief Data Scientist of IMIDomics, Inc. The remaining authors declare that they have no conflict of interest.
Figures



References
-
- Felson D.T., Smolen J.S., Wells G., et al. American College of Rheumatology/European League against Rheumatism provisional definition of remission in rheumatoid arthritis for clinical trials. Ann Rheum Dis. 2011;70(3):404–413. Mar. - PubMed
-
- Pitzalis C., Choy E.H., Buch M.H. Transforming clinical trials in rheumatology: towards patient-centric precision medicine. Nat Rev Rheumatol. 2020;16(10):590–599. - PubMed
-
- Taylor P.C., Feldmann M. Anti-TNF biologic agents: still the therapy of choice for rheumatoid arthritis. Nat Rev Rheumatol. 2009;5(10):578–582. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

