The Astrin-SKAP complex reduces friction at the kinetochore-microtubule interface
- PMID: 35580605
- PMCID: PMC9295892
- DOI: 10.1016/j.cub.2022.04.061
The Astrin-SKAP complex reduces friction at the kinetochore-microtubule interface
Abstract
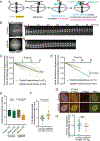
The kinetochore links chromosomes to spindle microtubules to drive chromosome segregation at cell division. While we know nearly all mammalian kinetochore proteins, how these give rise to the strong yet dynamic microtubule attachments required for function remains poorly understood. Here, we focus on the Astrin-SKAP complex, which localizes to bioriented kinetochores and is essential for chromosome segregation but whose mechanical role is unclear. Live imaging reveals that SKAP depletion dampens the movement and decreases the coordination of metaphase sister kinetochores and increases the tension between them. Using laser ablation to isolate kinetochores bound to polymerizing versus depolymerizing microtubules, we show that without SKAP, kinetochores move slower on both polymerizing and depolymerizing microtubules and that more force is needed to rescue microtubules to polymerize. Thus, in contrast to the previously described kinetochore proteins that increase the grip on microtubules under force, Astrin-SKAP reduces the grip, increasing attachment dynamics and force responsiveness and reducing friction. Together, our findings suggest a model where the Astrin-SKAP complex effectively "lubricates" correct, bioriented attachments to help preserve them.
Keywords: Astrin-SKAP; dynamics; force; friction; grip; kinetochore; mammal; mechanics; microtubule; spindle.
Copyright © 2022 Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests The authors declare no conflict of interest.
Figures




Comment in
-
Cell division: The science friction of chromosome attachment.Curr Biol. 2022 Jul 11;32(13):R744-R746. doi: 10.1016/j.cub.2022.05.045. Curr Biol. 2022. PMID: 35820385
Similar articles
-
SKAP binding to microtubules reduces friction at the kinetochore-microtubule interface and increases attachment stability under force.Curr Biol. 2025 Apr 21;35(8):1805-1815.e4. doi: 10.1016/j.cub.2025.03.003. Epub 2025 Mar 27. Curr Biol. 2025. PMID: 40154475
-
The association of Plk1 with the astrin-kinastrin complex promotes formation and maintenance of a metaphase plate.J Cell Sci. 2021 Jan 8;134(1):jcs251025. doi: 10.1242/jcs.251025. J Cell Sci. 2021. PMID: 33288550 Free PMC article.
-
Astrin-SKAP complex reconstitution reveals its kinetochore interaction with microtubule-bound Ndc80.Elife. 2017 Aug 25;6:e26866. doi: 10.7554/eLife.26866. Elife. 2017. PMID: 28841134 Free PMC article.
-
Merotelic kinetochores in mammalian tissue cells.Philos Trans R Soc Lond B Biol Sci. 2005 Mar 29;360(1455):553-68. doi: 10.1098/rstb.2004.1610. Philos Trans R Soc Lond B Biol Sci. 2005. PMID: 15897180 Free PMC article. Review.
-
The kinetochore-microtubule interface at a glance.J Cell Sci. 2018 Aug 16;131(16):jcs214577. doi: 10.1242/jcs.214577. J Cell Sci. 2018. PMID: 30115751 Free PMC article. Review.
Cited by
-
α-tubulin detyrosination fine-tunes kinetochore-microtubule attachments.Nat Commun. 2024 Nov 9;15(1):9720. doi: 10.1038/s41467-024-54155-8. Nat Commun. 2024. PMID: 39521805 Free PMC article.
-
Liquid-liquid phase separation of microtubule-binding proteins in the regulation of spindle assembly.Cell Prolif. 2024 Oct;57(10):e13649. doi: 10.1111/cpr.13649. Epub 2024 May 13. Cell Prolif. 2024. PMID: 38736355 Free PMC article. Review.
-
The Four Causes: The Functional Architecture of Centromeres and Kinetochores.Annu Rev Genet. 2022 Nov 30;56:279-314. doi: 10.1146/annurev-genet-072820-034559. Epub 2022 Sep 2. Annu Rev Genet. 2022. PMID: 36055650 Free PMC article. Review.
-
PP6 regulation of Aurora A-TPX2 limits NDC80 phosphorylation and mitotic spindle size.J Cell Biol. 2023 May 1;222(5):e202205117. doi: 10.1083/jcb.202205117. Epub 2023 Mar 10. J Cell Biol. 2023. PMID: 36897279 Free PMC article.
-
SKAP binding to microtubules reduces friction at the kinetochore-microtubule interface and increases attachment stability under force.bioRxiv [Preprint]. 2024 Aug 8:2024.08.08.607154. doi: 10.1101/2024.08.08.607154. bioRxiv. 2024. Update in: Curr Biol. 2025 Apr 21;35(8):1805-1815.e4. doi: 10.1016/j.cub.2025.03.003. PMID: 39149232 Free PMC article. Updated. Preprint.
References
-
- Wendell KL, Wilson L, and Jordan MA (1993). Mitotic block in HeLa cells by vinblastine: Ultrastructural changes in kinetochore-mcirotubule attachment and in centrosomes. J. Cell Sci 104, 261–274. - PubMed

