Ultrasound triggered organic mechanoluminescence materials
- PMID: 35580814
- PMCID: PMC10202817
- DOI: 10.1016/j.addr.2022.114343
Ultrasound triggered organic mechanoluminescence materials
Abstract
Ultrasound induced organic mechanoluminescence materials have become one of the focal topics in wireless light sources since they exhibit high spatiotemporal resolution, biocompatibility and excellent tissue penetration depth. These properties promote great potential in ultrahigh sensitive bioimaging with no background noise and noninvasive nanodevices. Recent advances in chemistry, nanotechnology and biomedical research are revolutionizing ultrasound induced organic mechanoluminescence. Herein, we try to summarize some recent researches in ultrasound induced mechanoluminescence that use various materials design strategies based on the molecular conformational changes and cycloreversion reaction. Practical applications, like noninvasive bioimaging and noninvasive optogenetics, are also presented and prospected.
Keywords: Acoustic cavitation; Bioimaging; Focused ultrasound; Mechanoluminescence; Photocatalysis; ROS; Sonochemistry.
Copyright © 2022 Elsevier B.V. All rights reserved.
Conflict of interest statement
Declaration of Competing Interest The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures



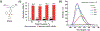

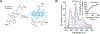
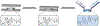
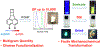
Similar articles
-
Principles and applications of sono-optogenetics.Adv Drug Deliv Rev. 2023 Mar;194:114711. doi: 10.1016/j.addr.2023.114711. Epub 2023 Jan 25. Adv Drug Deliv Rev. 2023. PMID: 36708773 Free PMC article. Review.
-
Mechanically Triggered Bright Chemiluminescence from Polymers by Exploiting a Synergy between Masked 2-Furylcarbinol Mechanophores and 1,2-Dioxetane Chemiluminophores.J Am Chem Soc. 2024 Aug 14;146(32):22151-22156. doi: 10.1021/jacs.4c07592. Epub 2024 Jul 30. J Am Chem Soc. 2024. PMID: 39078378 Free PMC article.
-
Ultrasound-Triggered In Situ Photon Emission for Noninvasive Optogenetics.J Am Chem Soc. 2023 Jan 18;145(2):1097-1107. doi: 10.1021/jacs.2c10666. Epub 2023 Jan 6. J Am Chem Soc. 2023. PMID: 36606703 Free PMC article.
-
Isostructural doping for organic persistent mechanoluminescence.Nat Commun. 2024 Apr 30;15(1):3668. doi: 10.1038/s41467-024-47962-6. Nat Commun. 2024. PMID: 38693122 Free PMC article.
-
Sonochemistry and sonoprocessing: the link, the trends and (probably) the future.Ultrason Sonochem. 2003 Jul;10(4-5):175-9. doi: 10.1016/S1350-4177(03)00086-5. Ultrason Sonochem. 2003. PMID: 12818379 Review.
Cited by
-
Principles and applications of sono-optogenetics.Adv Drug Deliv Rev. 2023 Mar;194:114711. doi: 10.1016/j.addr.2023.114711. Epub 2023 Jan 25. Adv Drug Deliv Rev. 2023. PMID: 36708773 Free PMC article. Review.
-
Hydrogels in wearable neural interfaces.Med X. 2024;2(1):23. doi: 10.1007/s44258-024-00040-4. Epub 2024 Dec 9. Med X. 2024. PMID: 39659711 Free PMC article. Review.
-
Explore Ultrasonic-Induced Mechanoluminescent Solutions towards Realising Remote Structural Health Monitoring.Sensors (Basel). 2024 Jul 16;24(14):4595. doi: 10.3390/s24144595. Sensors (Basel). 2024. PMID: 39065996 Free PMC article.
-
Mechanically Triggered Bright Chemiluminescence from Polymers by Exploiting a Synergy between Masked 2-Furylcarbinol Mechanophores and 1,2-Dioxetane Chemiluminophores.J Am Chem Soc. 2024 Aug 14;146(32):22151-22156. doi: 10.1021/jacs.4c07592. Epub 2024 Jul 30. J Am Chem Soc. 2024. PMID: 39078378 Free PMC article.
-
Ultrasound-Controlled Prodrug Activation: Emerging Strategies in Polymer Mechanochemistry and Sonodynamic Therapy.ACS Appl Bio Mater. 2024 Dec 16;7(12):8040-8058. doi: 10.1021/acsabm.4c00150. Epub 2024 May 2. ACS Appl Bio Mater. 2024. PMID: 38698527 Free PMC article. Review.
References
-
- Zhegunov G, Strategy of Matter and Energy Transformation, in: Zhegunov G (Ed.), The Dual Nature of Life: Interplay of the Individual and the Genome, Springer, Berlin Heidelberg, Berlin, Heidelberg, 2012, pp. 165–182.
-
- Hollwich F, The Influence of Ocular Light Perception on Metabolism in Man and in Animal, Springer, New York, 2011.
-
- Björn LO, Photoreversibly photochromic pigments in organisms: properties and role in biological light perception, Q. Rev. Biophys 12 (1979) 1–23. - PubMed
-
- Kricka LJ, Clinical applications of chemiluminescence, Anal. Chim. Acta 500 (1–2) (2003) 279–286.
-
- He L.i., Peng ZW, Jiang ZW, Tang XQ, Huang CZ, Li YF, Novel iron(III)-based metal–organic gels with superior catalytic performance toward luminol chemiluminescence, ACS Appl. Mater. Interfaces 9 (37) (2017) 31834–31840. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

