A double-blind, Randomized controlled trial on glucose-lowering EFfects and safety of adding 0.25 or 0.5 mg lobeglitazone in type 2 diabetes patients with INadequate control on metformin and dipeptidyl peptidase-4 inhibitor therapy: REFIND study
- PMID: 35581902
- PMCID: PMC9541308
- DOI: 10.1111/dom.14766
A double-blind, Randomized controlled trial on glucose-lowering EFfects and safety of adding 0.25 or 0.5 mg lobeglitazone in type 2 diabetes patients with INadequate control on metformin and dipeptidyl peptidase-4 inhibitor therapy: REFIND study
Abstract
Aims: To compare the efficacy and safety of adding low-dose lobeglitazone (0.25 mg/day) or standard-dose lobeglitazone (0.5 mg/day) to patients with type 2 diabetes mellitus (T2DM) with inadequate glucose control on metformin and dipeptidyl peptidase (DPP4) inhibitor therapy.
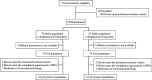
Materials and methods: In this phase 4, multicentre, double-blind, randomized controlled, non-inferiority trial, patients with T2DM insufficiently controlled by metformin and DPP4 inhibitor combination therapy were randomized to receive either low-dose or standard-dose lobeglitazone. The primary endpoint was non-inferiority of low-dose lobeglitazone in terms of glycaemic control, expressed as the difference in mean glycated haemoglobin levels at week 24 relative to baseline values and compared with standard-dose lobeglitazone, using 0.5% non-inferiority margin.
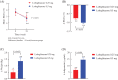
Results: At week 24, the mean glycated haemoglobin levels were 6.87 ± 0.54% and 6.68 ± 0.46% in low-dose and standard-dose lobeglitazone groups, respectively (p = .031). The between-group difference was 0.18% (95% confidence interval 0.017-0.345), showing non-inferiority of the low-dose lobeglitazone. Mean body weight changes were significantly greater in the standard-dose group (1.36 ± 2.23 kg) than in the low-dose group (0.50 ± 1.85 kg) at week 24. The changes in HOMA-IR, lipid profile and liver enzyme levels showed no significant difference between the groups. Overall treatment-emergent adverse events (including weight gain, oedema and hypoglycaemia) occurred more frequently in the standard-dose group.
Conclusions: Adding low-dose lobeglitazone to metformin and DPP4 inhibitor combination resulted in a non-inferior glucose-lowering outcome and fewer adverse events compared with standard-dose lobeglitazone. Therefore, low-dose lobeglitazone might be one option for individualized strategy in patients with T2DM.
Keywords: antidiabetic drugs; beta-cell function; glycaemic control; thiazolidinediones; type 2 diabetes.
© 2022 The Authors. Diabetes, Obesity and Metabolism published by John Wiley & Sons Ltd.
Conflict of interest statement
This study was supported by a research grant from Chong Kun Dang Pharmaceutical Corporation, Seoul, Republic of Korea. The authors declare that they have no competing interests.
Figures


References
-
- Aschner P, Karuranga S, James S, et al. The international diabetes Federation's guide for diabetes epidemiological studies. Diabetes Res Clin Pract. 2021;172:108630. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

