Calcium carbonate supplementation causes motor dysfunction
- PMID: 35584940
- PMCID: PMC9388334
- DOI: 10.1538/expanim.22-0011
Calcium carbonate supplementation causes motor dysfunction
Abstract
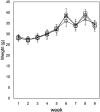
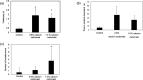
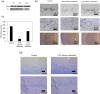
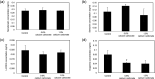
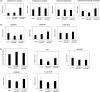
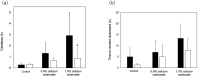
We previously showed that a diet containing calcium carbonate causes impairments in spatial and recognition memory in mice. In this study, we investigated the effects of calcium carbonate supplementation on motor function. Motor function was determined using different tests that have been used to analyze different aspects of Parkinsonism. A catalepsy test for akinesia; a muscular strength assessment, pole test, beam-walking test, and gait analysis for motor coordination and balance assessment; and an open-field test for locomotor activity assessment were performed. The mice were fed diets containing 0.6% or 1.0% calcium carbonate for eight weeks, after which they were evaluated for motor functions. The diets containing calcium carbonate caused significant motor dysfunction, as revealed by the different tests, although the spontaneous locomotor activity did not change. Calcium carbonate supplementation decreased the dopamine content in the basal ganglia, including the striatum and substantia nigra, and the number of tyrosine hydroxylase-positive neurons in the substantia nigra. In addition, administration of L-dopa led to at least a partial recovery of motor dysfunction, suggesting that calcium carbonate supplementation causes motor dysfunction by decreasing the dopamine content in the basal ganglia. These results suggest that mice with calcium carbonate-induced motor dysfunction may be useful as a new animal model for Parkinson's disease and Huntington's disease.
Keywords: Parkinson’s disease; calcium carbonate supplements; dopaminergic neurons; motor dysfunction.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Combined low calcium and lack magnesium is a risk factor for motor deficit in mice.Biosci Biotechnol Biochem. 2013;77(2):266-70. doi: 10.1271/bbb.120671. Epub 2013 Feb 7. Biosci Biotechnol Biochem. 2013. PMID: 23391905
-
Conditional deletion of Ndufs4 in dopaminergic neurons promotes Parkinson's disease-like non-motor symptoms without loss of dopamine neurons.Sci Rep. 2017 Mar 22;7:44989. doi: 10.1038/srep44989. Sci Rep. 2017. PMID: 28327638 Free PMC article.
-
The pesticide fipronil injected into the substantia nigra of male rats decreases striatal dopamine content: A neurochemical, immunohistochemical and behavioral study.Behav Brain Res. 2020 Apr 20;384:112562. doi: 10.1016/j.bbr.2020.112562. Epub 2020 Feb 15. Behav Brain Res. 2020. PMID: 32070689
-
Quantitative assessment of gait and neurochemical correlation in a classical murine model of Parkinson's disease.BMC Neurosci. 2012 Nov 14;13:142. doi: 10.1186/1471-2202-13-142. BMC Neurosci. 2012. PMID: 23151254 Free PMC article.
-
Chapter 33: the history of movement disorders.Handb Clin Neurol. 2010;95:501-46. doi: 10.1016/S0072-9752(08)02133-7. Handb Clin Neurol. 2010. PMID: 19892136 Review.
Cited by
-
Impaired therapeutic efficacy of sleep-deprived bone marrow mesenchymal stem cells in the cerebellum of mouse cuprizone model of multiple sclerosis: stereological and motor behavioral evidence.J Mol Histol. 2025 Jul 5;56(4):217. doi: 10.1007/s10735-025-10509-0. J Mol Histol. 2025. PMID: 40616615

