Attenuation of Excess TNF-α Release in Crohn's Disease by Silencing of iRHOMs 1/2 and the Restoration of TGF-β Mediated Immunosuppression Through Modulation of TACE Trafficking
- PMID: 35585977
- PMCID: PMC9108260
- DOI: 10.3389/fimmu.2022.887830
Attenuation of Excess TNF-α Release in Crohn's Disease by Silencing of iRHOMs 1/2 and the Restoration of TGF-β Mediated Immunosuppression Through Modulation of TACE Trafficking
Abstract
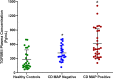
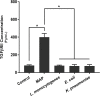
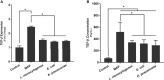
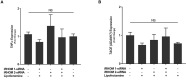
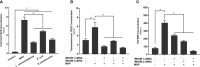
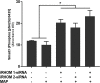
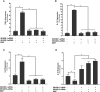
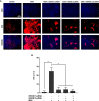
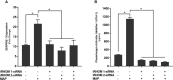
TNFα converting enzyme (TACE) is a transmembrane metalloprotease that sheds an assortment of signaling receptors, cytokines, growth factors, and pro-inflammatory mediators. In Crohn's disease (CD), TACE activity is upregulated, resulting in a marked increase of TNFα secretion and inflammation. Although treatment of CD with TNFα monoclonal antibodies is beneficial, many patients are at risk for acquiring opportunistic infections, and the treatment efficacy of TNFα monoclonal antibodies typically decreases over time. This study investigated an alternative approach for mitigating TNFα release by knocking down TACE membrane translocation in macrophages via inhibitory rhomboid proteins 1 and 2 (iRHOMs 1/2) siRNA treatment. First we measured TGFβRII shedding in ex vivo plasma samples collected from CD patients and healthy control subjects (N=40 per group). Then, we measured TGFβRII shedding and the expression and production of TGFβ ligand, TNFα, IL-6, IL-1β, IL-10, and total versus membranous TACE in vitro with THP-1 derived macrophage infected with Mycobacterium avium subspecies paratuberculosis (MAP), a highly studied CD-related pathogen. We determined that TGFβRII shedding was significantly higher in CD patients compared to healthy controls [515.52 ± 54.23 pg/mL vs 310.81 ± 43.16 pg/mL, respectively], and MAP-infected CD plasma samples had significantly more TGFβRII shedding (601.83 ± 49.56 pg/mL) than MAP-negative CD samples (430.37 ± 45.73 pg/mL). Moreover, we also determined that TACE production; TGFβ ligand expression and production; and TGFβRII shedding were also higher in MAP-infected THP-1 macrophages. Nevertheless, once we transfected the MAP infected macrophages with iRHOM siRNA, TACE production and membrane localization were significantly decreased, resulting in a significant decrease in TGFβRII shedding; an increase in Smad3 phosphorylation; a decrease in the expression and production of pro-inflammatory cytokines; and a decrease in the expression and production of stricture-associated factor, plasminogen activator inhibitor-1 (PAI-1). Our data clearly demonstrates that the regression of TACE trafficking, via iRHOM 1/2 silencing, significantly reduces the release of TNFα and restores the immunosuppressive capabilities of TGFβ signaling, which ultimately reverses inflammatory tissue damage. Accordingly, this study may provide a framework for the creation of newer, safer therapeutic options designed to treat inflammatory autoimmune diseases such as CD and rheumatoid arthritis.
Keywords: Crohn's disease; TACE (TNF-α converting enzyme); ectodomain shedding; iRHOM 1/2; mycobacteria paratuberculosis; rhomboid proteins.
Copyright © 2022 Louis, Qasem and Naser.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

