Prophylactic antiemetics for adults receiving intravenous opioids in the acute care setting
- PMID: 35588093
- PMCID: PMC9119423
- DOI: 10.1002/14651858.CD013860.pub2
Prophylactic antiemetics for adults receiving intravenous opioids in the acute care setting
Abstract
Background: Physicians often prescribe opioids for pain in the acute care setting. Nausea and vomiting are well-described adverse events, occurring in over one-third of patients. Prophylactic antiemetics may be one option to reduce opioid-associated nausea and vomiting. However, these medications also have their own adverse effects, so it is important to understand their efficacy and safety prior to routine use. This is a review of randomized controlled trials comparing prophylactic antiemetics versus placebo or standard care for preventing opioid-associated nausea and vomiting.
Objectives: To assess the effects of prophylactic antiemetics for nausea and vomiting in adults (aged 16 years or older) receiving intravenous opioids in the acute care setting.
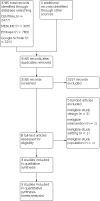
Search methods: We searched CENTRAL (the Cochrane Library), MEDLINE (OVID), Embase (OVID) from inception to January 2022, and Google Scholar (17 January 2022). We also searched the World Health Organization (WHO) International Clinical Trials Registry Platform (ICTRP) and screened reference lists.
Selection criteria: We included randomized controlled trials of prophylactic antiemetics versus placebo or standard care in adults prior to receiving an intravenous opioid.
Data collection and analysis: Two review authors (MG, JNC) independently determined the eligibility of each study according to the inclusion criteria. Two review authors (MG, GDP) then independently extracted data, assessed risk of bias, and determined the certainty of evidence using GRADE. Our primary outcomes were the occurrence of nausea, vomiting, and adverse events. Secondary outcomes included nausea severity, number of vomiting episodes, and number of participants requiring antiemetic rescue therapy. We presented outcomes as risk ratios (RR) for dichotomous data (e.g. presence of vomiting, presence of nausea, number of participants requiring rescue medication, adverse events) and mean difference (MD) or standardized mean difference for continuous data (e.g. number of vomiting episodes, nausea severity) with 95% confidence intervals (CI).
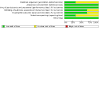
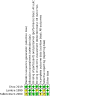
Main results: We included three studies involving 527 participants (187 women and 340 men) with a mean age of 42 years. All studies used intravenous metoclopramide (10 mg) as the intervention and a placebo for the comparator. No studies assessed any other antiemetic or compared the intervention to standard care. Compared to placebo, metoclopramide did not reduce vomiting (RR 1.18, 95% CI 0.26 to 5.32; low-certainty evidence) or nausea (RR 0.55; 95% CI 0.15 to 2.03; low-certainty evidence) and there was no difference in adverse events (RR 2.34, 95% CI 0.47 to 11.61; low-certainty evidence). No data were available regarding the number of vomiting episodes. Metoclopramide did reduce the severity of nausea compared with placebo (MD -0.49, 95% CI -0.75 to -0.23; low-certainty evidence) but did not reduce the need for rescue medication (RR 1.86, 95% CI 0.17 to 20.16; low-certainty evidence). Two studies were at unclear risk of bias for random sequence generation, one for blinding of outcome assessors, one for incomplete outcome data, and two for selective reporting. The studies were at low risk of bias for all remaining components.
Authors' conclusions: There was no evidence that prophylactic metoclopramide affected the risk of vomiting, nausea, or the need for rescue medication when provided prior to intravenous opioids in the acute care setting. There was a clinically insignificant difference in nausea severity when comparing prophylactic metoclopramide with placebo. Overall, the evidence was of low certainty. Future research could better delineate the effects of prophylactic antiemetics on specific populations, and new studies are needed to evaluate the use of other prophylactic antiemetic agents, for which there were no data.
Copyright © 2022 The Cochrane Collaboration. Published by John Wiley & Sons, Ltd.
Conflict of interest statement
MG: none known. MG is an emergency medicine physician who manages pain in the acute care setting.
JNC: none known. JNC is an emergency medicine physician who manages pain in the acute care setting.
GDP: none known. GDP is a clinical pharmacist who works in the Emergency Department.
Figures












Update of
- doi: 10.1002/14651858.CD013860
Similar articles
-
Olanzapine for the prevention and treatment of cancer-related nausea and vomiting in adults.Cochrane Database Syst Rev. 2018 Sep 21;9(9):CD012555. doi: 10.1002/14651858.CD012555.pub2. Cochrane Database Syst Rev. 2018. PMID: 30246876 Free PMC article.
-
Aromatherapy for treatment of postoperative nausea and vomiting.Cochrane Database Syst Rev. 2018 Mar 10;3(3):CD007598. doi: 10.1002/14651858.CD007598.pub3. Cochrane Database Syst Rev. 2018. PMID: 29523018 Free PMC article.
-
Oxycodone for cancer-related pain.Cochrane Database Syst Rev. 2022 Jun 9;6(6):CD003870. doi: 10.1002/14651858.CD003870.pub7. Cochrane Database Syst Rev. 2022. PMID: 35679121 Free PMC article.
-
Antihistamines for motion sickness.Cochrane Database Syst Rev. 2022 Oct 17;10(10):CD012715. doi: 10.1002/14651858.CD012715.pub2. Cochrane Database Syst Rev. 2022. PMID: 36250781 Free PMC article.
-
Continuous intravenous perioperative lidocaine infusion for postoperative pain and recovery in adults.Cochrane Database Syst Rev. 2018 Jun 4;6(6):CD009642. doi: 10.1002/14651858.CD009642.pub3. Cochrane Database Syst Rev. 2018. PMID: 29864216 Free PMC article.
Cited by
-
The efficacy of prophylactic metoclopramide in preventing nausea and vomiting in patients with acute pain treated with intravenous tramadol: a randomized double-blinded, placebo-controlled trial.BMC Res Notes. 2023 Jul 27;16(1):158. doi: 10.1186/s13104-023-06395-y. BMC Res Notes. 2023. PMID: 37501098 Free PMC article. Clinical Trial.
References
References to studies included in this review
Choo 2019 {published data only}
-
- Choo KH, Manikam RA, Yoong KPY, Kandasamy VA.Prophylactic metoclopramide use in trauma patients given tramadol: a randomised, double-blinded, placebo-controlled trial [ ]. Hong Kong Journal of Emergency Medicine 2019;26(2):98-105.
Lambie 1999 {published data only}
-
- Lambie B, Chambers J, Herbison P.The role of prophylactic anti‐emetic therapy in emergency department patients receiving intravenous morphine for musculoskeletal trauma. Emergency Medicine Australasia 1999;11(4):240-3.
Talbot‐Stern 2000 {published data only}
-
- Talbot-Stern J, Paoloni R.Prophylactic metoclopramide is unnecessary with intravenous analgesia in the ED. American Journal of Emergency Medicine 2000;18(6):653-7. - PubMed
References to studies excluded from this review
Bradshaw 2006 {published data only}
Conner 1977 {published data only}
-
- Conner JT, Bellville JW, Wender R, Wapner S, Dorey FJ, Katz RL.Morphine and promethazine as intravenous premedicants. Anesthesia & Analgesia 1977;56(6):801-7. - PubMed
Culver 2017 {published data only}
-
- Culver MA, Richards EC, Jarrell DH, Edwards CJ.Use of prophylactic ondansetron with intravenous opioids in emergency department patients: a prospective observational pilot study. Journal of Emergency Medicine 2017;53(5):629-34. - PubMed
Hersh 2016 {published data only}
-
- Hersh E, Marino M, Schachtel B.CL-108 reduces the use of rescue antiemetic medications for opioid-induced nausea and vomiting. Pain Medicine 2018;19(4):423-4.
Okamoto 2007 {published data only}
-
- Okamoto Y, Tsuneto S, Matsuda Y, Inoue T, Tanimukai H, Tazumi K, et al.A retrospective chart review of the antiemetic effectiveness of risperidone in refractory opioid-induced nausea and vomiting in advanced cancer patients. Journal of Pain and Symptom Management 2007;34(2):217-22. - PubMed
Additional references
Bakhsh 2019
-
- Bakhsh HT, Perona SJ.Medical and nursing staff education reduces use of prophylactic ondansetron with opioids in the emergency department. Journal of Emergency Nursing 2019;45(3):273-7. - PubMed
Bramer 2017
Carlisle 2006
Chang 2014
-
- Chang HY, Daubresse M, Kruszewski SP, Alexander GC.Prevalence and treatment of pain in EDs in the United States, 2000 to 2010. American Journal of Emergency Medicine 2014;32(5):421-31. - PubMed
Coluzzi 2012
-
- Coluzzi F, Rocco A, Mandatori I, Mattia C.Non-analgesic effects of opioids: opioid-induced nausea and vomiting: mechanisms and strategies for their limitation. Current Pharmaceutical Design 2012;18(37):6043-52. - PubMed
Deeks 2020
-
- Deeks JJ, Higgins JP, Altman DG, editor(s).Chapter 10: Analysing data and undertaking meta-analyses. In: Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 6.1 (updated September 2020). Cochrane, 2020 [ ]. Available from training.cochrane.org/handbook ; ( ): .
Diemunsch 2009
-
- Diemunsch P, Joshi GP, Brichant JF.Neurokinin-1 receptor antagonists in the prevention of postoperative nausea and vomiting. British Journal of Anaesthesia 2009;103(1):7-13. - PubMed
Egger 1997
Elbourne 2002
-
- Elbourne DR, Altman DG, Higgins JP, Curtin F, Worthington HV, Vail A .Meta-analyses involving cross-over trials: methodological issues. International Journal of Epidemiology 2002;31(1):140-9. - PubMed
Friedman 2016
Fry 2011
Gan 2020
-
- Gan TJ, Belani KG, Bergese S, Chung F, Diemunsch P, Habib AS, et al.Fourth consensus guidelines for the management of postoperative nausea and vomiting. Anesthesia & Analgesia 2020;131(2):411-48. - PubMed
GRADEpro GDT 2015 [Computer program]
-
- McMaster University (developed by Evidence Prime, Inc.) GRADEpro Guideline Development Tool[ ]. , Version accessed prior to 13 April 2022. Hamilton (ON): McMaster University (developed by Evidence Prime, Inc.), . Available from www.gradepro.org.
Higgins 2011
-
- Higgins JP, Altman DG, Sterne JA.Chapter 8: Assessing risk of bias in included studies. In: Higgins JP, Green S, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from training.cochrane.org/handbook/archive/v5.1.
Higgins 2020
-
- Higgins JP, Eldridge S, Li T.Chapter 23: Including variants on randomized trials. In: Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA, editor(s). Cochrane Handbook for Systematic Reviews of Interventions version 6.1 (updated September 2020). Cochrane, 2020. Available from www.training.cochrane.org/handbook.
Johnston 1998
-
- Johnston CC, Gagnon AJ, Fullerton L, CommonC, Ladores M, Forlini S.One-week survey of pain intensity on admission to and discharge from the emergency department: a pilot study. Journal of Emergency Medicine 1998;16(3):377-82. - PubMed
Leow 2006
-
- Leow F, Knott J, Abu Hassan F, Taylor D, Udayasiri R.Risk and severity of akathisia following administration of metoclopramide in the emergency department. Journal of Pharmacy Practice and Research 2006;36:194-8.
Lesniak 2011
-
- Lesniak A, Lipkowski AW.Opioid peptides in peripheral pain control. Acta Neurobiologiae Experimentalis Journal 2011;71(1):129-38. - PubMed
Macario 1999
-
- Macario A, Weinger M, Carney S, Kim A.Which clinical anesthesia outcomes are important to avoid? The perspective of patients. Anesthesia and Analgesia 1999;89(3):652-8. - PubMed
Mallick‐Searle 2017
-
- Mallick-Searle T, Fillman M.The pathophysiology, incidence, impact, and treatment of opioid-induced nausea and vomiting. Journal of the American Association of Nurse Practitioners 2017;29(11):704-10. - PubMed
Mansour 1994
Meek 2009
-
- Meek R, Kelly AM, Hu XF.Use of the visual analog scale to rate and monitor severity of nausea in the emergency department. Academic Emergency Medicine 2009;16(12):1304-10. - PubMed
Moher 2009
Mura 2017
Parlak 2005
Rainer 2000
Review Manager 2020 [Computer program]
-
- Nordic Cochrane Centre, The Cochrane Collaboration Review Manager 5 (RevMan 5)[ ]. , Version 5.4. Copenhagen: Nordic Cochrane Centre, The Cochrane Collaboration, 2020. .
Roila 1995
-
- Roila F, Del Favero A.Ondansetron clinical pharmacokinetics. Clinical Pharmacokinetics 1995;29(2):95-109. - PubMed
Rui 2019
-
- Rui P, Schappert SM.Opioids prescribed at discharge or given during emergency department visits among adults in the United States, 2016 [ ]. US Department of Health and Human Services, Centers for Disease Control and Prevention; 2019 May. NCHS Data Brief. No 338. Available at: www.cdc.gov/nchs/data/databriefs/db338-h.pdf ; ( ): . - PubMed
Schünemann 2013
-
- Schünemann H, Brożek J, Guyatt G, Oxman A.Handbook for grading the quality of evidence and the strength of recommendations using the GRADE approach (updated October 2013). GRADE Working Group, 2013 [ ]. Available from gdt.guidelinedevelopment.org/app/handbook/handbook.html ; ( ): .
Schünemann 2020
-
- Schünemann HJ, Higgins JP, Vist GE, Glasziou P, Akl EA, Skoetz N, et al.Chapter 14: Completing ‘Summary of findings’ tables and grading the certainty of the evidence. In: Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA, editor(s). Cochrane Handbook for Systematic Reviews of Interventions version 6.1 (updated September 2020). Cochrane, 2020. Available from www.training.cochrane.org/handbook.
Simpson 2011
-
- Simpson PM, Bendall JC, Middleton PM.Review article: prophylactic metoclopramide for patients receiving intravenous morphine in the emergency setting: a systematic review and meta-analysis of randomized controlled trials [ ]. Emergency Medicine Australasia 2011;23(4):452-7. - PubMed
Tanabe 1999
-
- Tanabe P, Buschmann M.A prospective study of ED pain management practices and the patient's perspective. Journal of Emergency Nursing 1999;25(3):171-7. - PubMed
Weibel 2020
-
- Weibel S, Rücker G, Eberhart LH, Pace NL, Hartl HM, Jordan OL, et al.Drugs for preventing postoperative nausea and vomiting in adults after general anaesthesia: a network meta-analysis. Cochrane Database of Systematic Reviews 2020, Issue 10. Art. No: CD012859. [DOI: 10.1002/14651858.CD012859.pub2] - DOI - PMC - PubMed
Yeoh 2009
-
- Yeoh B, Taylor D, Taylor S.Education initiative improves the evidence-based use of metoclopramide following morphine administration in the emergency department. Emergency Medicine Australasia 2009;21:178-83. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

