Activation of the transcription factor NRF2 mediates the anti-inflammatory properties of a subset of over-the-counter and prescription NSAIDs
- PMID: 35588739
- PMCID: PMC9205175
- DOI: 10.1016/j.immuni.2022.04.015
Activation of the transcription factor NRF2 mediates the anti-inflammatory properties of a subset of over-the-counter and prescription NSAIDs
Abstract
Nonsteroidal anti-inflammatory drugs (NSAIDs) inhibit cyclooxygenase (COX) enzymes and are ubiquitously used for their anti-inflammatory properties. However, COX inhibition alone fails to explain numerous clinical outcomes of NSAID usage. Screening commonly used NSAIDs in primary human and murine myeloid cells demonstrated that NSAIDs could be differentiated by their ability to induce growth/differentiation factor 15 (GDF15), independent of COX specificity. Using genetic and pharmacologic approaches, NSAID-mediated GDF15 induction was dependent on the activation of nuclear factor erythroid 2-related factor 2 (NRF2) in myeloid cells. Sensing by Cysteine 151 of the NRF2 chaperone, Kelch-like ECH-associated protein 1 (KEAP1) was required for NSAID activation of NRF2 and subsequent anti-inflammatory effects both in vitro and in vivo. Myeloid-specific deletion of NRF2 abolished NSAID-mediated tissue protection in murine models of gout and endotoxemia. This highlights a noncanonical NRF2-dependent mechanism of action for the anti-inflammatory activity of a subset of commonly used NSAIDs.
Keywords: growth/differentiation factor 15; nonsteroidal anti-inflammatory drugs; nuclear factor erythroid 2-related factor 2.
Copyright © 2022 Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests P.T., H.T., and H.H.L. were employees of NGM Biopharmaceuticals at the time this work was done and may hold stock or stock options in this company. All data are available in the main text or the supplementary materials.
Figures
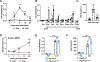

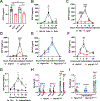


Comment in
-
Drugging inflammation: Easier NSAID than done.Immunity. 2022 Jun 14;55(6):973-975. doi: 10.1016/j.immuni.2022.05.009. Immunity. 2022. PMID: 35704999
References
-
- Al-Harbi NO, Nadeem A, Ahmad SF, AlThagfan SS, Alqinyah M, Alqahtani F, Ibrahim KE, and Al-Harbi MM (2019). Sulforaphane treatment reverses corticosteroid resistance in a mixed granulocytic mouse model of asthma by upregulation of antioxidants and attenuation of Th17 immune responses in the airways. Eur J Pharmacol 855, 276–284. - PubMed
-
- Angel J, Berenbaum F, Le Denmat C, Nevalainen T, Masliah J, and Fournier C (1994). Interleukin-1-induced prostaglandin E2 biosynthesis in human synovial cells involves the activation of cytosolic phospholipase A2 and cyclooxygenase-2. Eur J Biochem 226, 125–131. - PubMed
-
- Arias-Negrete S, Keller K, and Chadee K (1995). Proinflammatory cytokines regulate cyclooxygenase-2 mRNA expression in human macrophages. Biochem Biophys Res Commun 208, 582–589. - PubMed
-
- Baek SJ, Kim KS, Nixon JB, Wilson LC, and Eling TE (2001). Cyclooxygenase inhibitors regulate the expression of a TGF-beta superfamily member that has proapoptotic and antitumorigenic activities. Mol Pharmacol 59, 901–908. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

