Target receptor identification and subsequent treatment of resected brain tumors with encapsulated and engineered allogeneic stem cells
- PMID: 35589724
- PMCID: PMC9120173
- DOI: 10.1038/s41467-022-30558-3
Target receptor identification and subsequent treatment of resected brain tumors with encapsulated and engineered allogeneic stem cells
Retraction in
-
Retraction Note: Target receptor identification and subsequent treatment of resected brain tumors with encapsulated and engineered allogeneic stem cells.Nat Commun. 2025 Mar 21;16(1):2803. doi: 10.1038/s41467-025-57909-0. Nat Commun. 2025. PMID: 40118851 Free PMC article. No abstract available.
Abstract
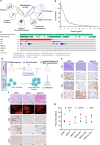
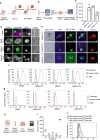
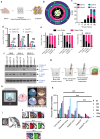
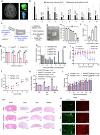
Cellular therapies offer a promising therapeutic strategy for the highly malignant brain tumor, glioblastoma (GBM). However, their clinical translation is limited by the lack of effective target identification and stringent testing in pre-clinical models that replicate standard treatment in GBM patients. In this study, we show the detection of cell surface death receptor (DR) target on CD146-enriched circulating tumor cells (CTC) captured from the blood of mice bearing GBM and patients diagnosed with GBM. Next, we developed allogeneic "off-the-shelf" clinical-grade bifunctional mesenchymal stem cells (MSCBif) expressing DR-targeted ligand and a safety kill switch. We show that biodegradable hydrogel encapsulated MSCBif (EnMSCBif) has a profound therapeutic efficacy in mice bearing patient-derived invasive, primary and recurrent GBM tumors following surgical resection. Activation of the kill switch enhances the efficacy of MSCBif and results in their elimination post-tumor treatment which can be tracked by positron emission tomography (PET) imaging. This study establishes a foundation towards a clinical trial of EnMSCBif in primary and recurrent GBM patients.
© 2022. The Author(s).
Conflict of interest statement
K.S. owns equity in and is a member of the Board of Directors of AMASA Therapeutics, a company developing stem cell-based therapies for cancer. D.B. owns equity in and is a consultant at AMASA Therapeutics. K.S.’s and D.B.’s interests were reviewed by Brigham and Women’s Hospital and Mass General Brigham in accordance with their conflict-of-interest policies. The other authors declare that they have no competing interests.
Figures

References
-
- Chmielewska, M., Losiewicz, K., Socha, P., Mecik-Kronenberg, T. & Wasowicz, K. The application of circulating tumor cells detecting methods in veterinary oncology. Pol. J. Vet. Sci.16, 141–151 (2013). - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

